Background
Epizootic ulcerative syndrome was defined at the DFID Regional Seminar on EUS in Bangkok in 1994 as "a seasonal epizootic condition of freshwater and estuarine warm water fish of complex infectious aetiology characterised by the presence of invasive Aphanomyces infection and necrotising ulcerative lesions typically leading to a granulomatous response" (Roberts et al., 1994). Research requirements identified from the meeting included work to compare and speciate fungal isolates from EUS outbreaks, develop diagnostic tests, and study the factors that affect transmission of the disease. Attempts are being made to apply DNA-based techniques to each of these study recommendations, and these are discussed here.
Molecular characterisation of EUS-associated Aphanomyces isolates
Traditionally, Oomycete fungal isolates are speciated primarily on the basis of the morphology of sexual structures. However many strains, and the more pathogenic strains in particular (including Aphanomyces astaci and Saprolegnia parasitica), are reluctant to produce sexual structures in culture. Sexual structures have not been demonstrated for the EUS Aphanomyces pathogen, and therefore alternative methods of characterisation have been used (Hatai and Egusa, 1978; Willoughby et al., 1995; Callinan et al., 1995; Lilley, 1997). Recently, DNA-based methods have also been applied.
Molecular data sets are rapidly becoming an essential part of any detailed fungal taxonomic study. Previous studies on other Aphanomyces and Saprolegnia species have used restriction fragment length polymorphism (RFLP) analyses to demonstrate inter-specific relationships (Yeh, 1989; Molina et al., 1995) and random amplification of polymorphic DNA (RAPD) studies have been used to show detailed intra-specific lineages (Huang et al., 1994; Malvick et al., 1998).
In studies comparing EUS-Aphanomyces isolates with isolates from mycotic granulomatosis (MG) and red spot disease (RSD) outbreaks, Hart (1997) analysed the ITS1-ITS4 region of the rRNA gene cluster using 10 enzymes (Alu I, Dde I, Hae III, Hha I, Hinf I, Hpa II, Hsp92 II, Mbo I, Rsa I and Sau96 I); and sequenced the NS5-NS6 and ITS1-ITS2 regions (Fig. 1). The isolates had all previously been shown to be slow-growing and pathogenic to snakehead fish when injected intramuscularly (Lilley and Roberts, 1997). These studies found no differences between any of the EUS, MG and RSD isolates.
A variety of saprophytic Aphanomyces, Achlya and Saprolegnia species isolated from the surface of EUS-affected fish or from infected waters, and Oomycete fungi involved in other diseases of aquatic animals, were also included in these analyses. These isolates had previously been shown to have very different temperature-growth profiles from the pathogens, and were incapable of growth within snakehead fish (Lilley and Roberts, 1997). The rRNA gene studies succeeded in differentiating all of these isolates from the EUS, MG and RSD pathogens (Hart, 1997). Dendrograms constructed from the RFLP data showed that the Aphanomyces pathogens clustered most closely to European isolates of the crayfish plague fungus, Aphanomyces astaci.
Lilley et al. (1997) used RAPD-PCR of genomic DNA to investigate possible intra-specific differences between the isolates. Twenty pathogenic isolates from several localities in Bangladesh, Thailand, Indonesia, Philippines, Australia and Japan were compared using 14 ten-mer primers (Fig 2). Also included in the study were 6 Aphanomyces saprophytes, 4 A. astaci isolates, and 2 Aphanomyces isolates from fish affected by ulcerative mycosis (UM) off the eastern coast of the USA. A total of 321 bands were used for the analysis. The mean similarity (F ± SD) between all the pathogens was calculated at 0.95 ± 0.03, whereas the other Aphanomyces species had a mean similarity of only 0.14 ± 0.05 compared with the pathogens. These results show that the EUS, MG and RSD pathogens are not only con-specific (now listed in the Index of Fungi as Aphanomyces invadans), but also genetically very similar. This indicates that the isolates are not long-term residents in each locality, but have spread across Asia relatively recently. In comparison, RAPD studies on A. astaci yielded four distinguishable groups from 15 European isolates indicating that there have been several introductions of that fungus to Europe over a number of years (Huang et al., 1994).
Figure 1. Structure of the
rRNA gene cluster and positions of fungal PCR primers. The cluster is split into
coding (18S, 5.8S and 28S genes) and non-coding (Internally Transcribed Spacer
or ITS) regions. The positions of the PCR primers and their direction of
synthesis are indicated by arrows.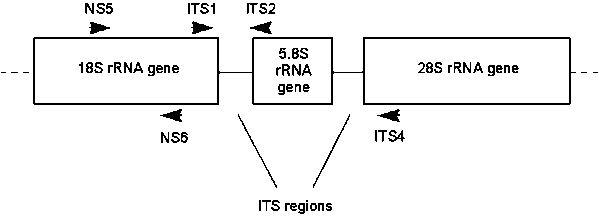
Figure 2. Sequence of 14 random 10-mer Operon primers used for RAPD analyses
A3 5'-AGTCAGCCAC
A4 5'-AATCGGGCTG
A6 5'-GGTCCCTGAC
A7 5'-GAAACGGGTG
A10 5'-GTGATCGCAG
A12 5'-TCGGCGATAG
A18 5'-AGGTGACCGT
A19 5'-CAAACGTCGG
A20 5'-GTTGCGATCC
B1 5'-GTTTCGCTCC
B2 5'-TGATCCCTGG
B4 5'-GGACTGGAGT
B5 5'-TGCGCCCTTC
B10 5'-CTGCTGGGAC
Diagnosis of EUS
Ulcerated fish are diagnosed as EUS-positive by histological demonstration of distinctive mycotic granulomas in underlying tissues. This is a reliable technique that yields a lot of information about the disease. As a result of the molecular characterisation work described above, a DNA probe for the specific detection of Aphanomyces invadans has been developed, and this could be used in a PCR-based diagnosis of EUS. However, the results of such a test would not give any information on the extent of infection, if indeed the fish is infected and not just carrying propagules of the fungus, or retaining fungal DNA from a past infection. PCR diagnoses also can suffer from problems of reliability and reproducibility, and in most EUS-affected areas it is a more expensive procedure than histology. Therefore, instead of PCR, attempts are being made to develop an in situ hybridisation technique using the probe. It is hoped this will enable histological sections to be further processed for the specific detection of A. invadans, and would compliment, rather than replace, histological diagnosis. The development and application of DNA probes for other agents associated with EUS (e.g. rhabdoviruses) would also provide further information about the involvement of these agents in EUS outbreaks.
PCR-based method to detect Aphanomyces invadans in the environment
To date, isolates of A. invadans have only been obtained from internal tissues of EUS-affected fish. Efforts have been made to recover A. invadans from natural water bodies in Thailand, but these have not succeeded due to colonisation of isolation media or fungus baits by faster-growing saprophytic fungi (Willoughby and Lilley, 1992). Fraser and Callinan (1996) used particular growth characteristics of A. invadans to devise a technique that excludes opportunist fungi. They were able to quantify A. invadans-like colonies on two occasions, but it has proved difficult to reproduce this technique reliably. As a result, important aspects of the natural ecology of A. invadans (e.g. persistence of the fungus in ponds outwith the EUS season, and fungus viability on resistant/carrier fish or on non-fish substrates) have yet to be studied.
Molecular detection techniques have been used to assay for Oomycete plant pathogens in environmental samples (Judelson and Messenger-Routh, 1996; Coelho et al., 1997; Liew et al., 1998) and DNA probes are presently being developed by researchers studying the toxin-producing dinoflagellate, Pfiesteria (Greer et al., 1997). Ulcerative disease outbreaks in eastern USA have been associated with an invasive mycosis as well as with Pfiesteria toxins (Blazer et al., 1998). In these cases, molecular techniques may be useful in detecting the various agents and determining their role in each outbreak.
A molecular assay technique has recently been devised to test for the presence of A. invadans DNA in water samples and other substrates. This is based on the PCR amplification of an A. invadans-specific 98 bp sequence, that was identified during the RFLP work described above. The particular problems of validating a PCR-based proxy detection method were identified by Hiney and Smith (1998) with regards to bacterial fish pathogens. They devised a study framework that evaluated quantitative, qualitative and reliability criteria at 4 levels of experimental complexity: (a) in vitro; (b) using a sterile seeded microcosm; (c) in a non-sterile incurred mesocosm; and (d) in non-sterile field samples. This approach can be applied to the study of A. invadans propagules in the environment. The planned study levels for this work are listed below.
a) The in vitro study aims to assess the specificity and sensitivity (DNA low/high detection limits) of the PCR assay on DNA extracted from cultured fungal mycelium. A range of A. invadans isolates from different localities is being studied against a strain panel of related species recovered from EUS-affected areas.
b) Sterile seeded mesocosm. A procedure is being developed to test the DNA extracted from fungal zoospores suspended in flasks of sterile distilled water. A. invadans spores will be tested among spores of fungi from the strain panel. Zoospore detection limits will be assessed by making haemocytometer counts of the zoospores present.
c) Non-sterile incurred mesocosm. Fungal zoospore suspensions will be induced in tanks containing pond water. Zoospore detection limits cannot be accurately assessed at this level as fungal species cannot be identified during haemocytometer zoospore counts. It is hoped the tank study can be used to assess the effect of different variables on the persistence of fungal DNA in the water (e.g. water quality, presence/absence of fish and other potential substrates, different treatment regimes).
d) The study of non-sterile field samples aims to test for A. invadans DNA in affected areas during non-outbreak periods, and in unaffected areas.
Conclusions
PCR-based techniques have provided valuable information in the study of the fungal pathogen involved in EUS outbreaks. In particular, research on the characterisation of A. invadans isolates has benefited from the use of these techniques, and it is envisaged that information on the ecology of the fungus will also be obtained with the use of PCR. It is hoped that the outputs from these latter studies will enable risk factors for the disease to be identified, so that recommendations can be made regarding control methods. It is not envisaged that PCR will be used in the routine diagnosis of EUS, but DNA-based methods may compliment histological diagnoses by providing A. invadans-specific stains using in situ hybridisation.
References
Blazer, V., Phillips, S. and Pendleton, E. (1998). Fish health, fungal infections, and Pfiesteria: The role of the U.S. Geological Survey. [WWW document] U.S. Geological Survey. (Last updated: Thursday 1 October 1998 08:07:34 MDT). http://biology.usgs.gov/pr/newsrelease/1998/9-23d.html.
Callinan, R.B., Paclibare, J.O., Bondad-Reantaso, M.G., Chin, J.C. and Gogolewski, R.P. (1995). Aphanomyces species associated with epizootic ulcerative syndrome (EUS) in the Philippines and red spot disease (RSD) in Australia: preliminary comparative studies. Diseases of Aquatic Organisms 21(3), 233-238.
Coelho, A.C., Cravador, A., Bollen, A., Ferraz, J.F.P., Moreira, A.C., Fauconnier, A. and Godfroid, E. (1997). Highly specific and sensitive non-radioactive molecular identification of Phytophthora cinnamomi. Mycological Research 101, 1499-1507.
Fraser, G. and Callinan, R. (1996). A technique for enumeration of propagules of the epizootic ulcerative syndrome fungus in pondwater. World Aquaculture 1996 Book of Abstracts. Queen Sirikit National Convention Centre, Bangkok, 29 January - 2 February 1996. P.131. The World Aquaculture Society.
Greer, J., Leffler, M., Belas, R., Kramer, J. and Place, A. (1997). In: Zohar, Y., Belas, R., Vasta, G., Place, A., and Kramer, J. (Organizing Committee), Molecular Technologies and Pfiesteria Research: A Scientific Synthesis. Report of a workshop held at the Center of Marine Biotechnology, University of Maryland Biotechnology Institute, 28-30 October 1997. A Maryland Sea Grant Publication, College Park, Maryland. http://www.mdsg.umd.edu/MDSG/
Hart, D. (1997). Development of diagnostic systems for the study of the molecular epidemiology and taxonomy of the Aphanomyces fungus associated with EUS of tropical and freshwater fishes. Final Report of DFID Research Project R5902Cb. 1st October 1993 to 31st March 1997.
Hatai, K. and Egusa, S. (1978). Studies on the pathogenic fungus of mycotic granulomatosis - II. Some of the note on the MG-fungus. Fish Pathology 13, 85-89. (In Japanese, English abstract).
Hiney, M.P. and Smith, P.R. (1998). Validation of polymerase chain reaction-based techniques for proxy detection of bacterial fish pathogens: Framework, problems and possible solutions for environmental applications. Aquaculture 162, 41-68.
Huang, T.-S., Cerenius, L. and Söderhäll, K. (1994). Analysis of genetic diversity in the crayfish plague fungus, Aphanomyces astaci, by random amplification of polymorphic DNA. Aquaculture 126, 1-10.
Judelson, H.S. and Messenger-Routh, B. (1996). Quantitation of Phytophthora cinnamomi in avocado roots using a species-specific DNA probe. Phytopathology 86, 763-768.
Liew, E.C.Y., Maclean, D.J. and Irwin, J.A.G. (1998). Specific PCR based detection of Phytophthora medicaginis using the intergenic spacer region of the ribosomal DNA. Mycological Research 102, 73-80.
Lilley, J. H. (1997). Studies on the comparative biology of Aphanomyces invadans. PhD thesis, University of Stirling, Scotland. 228 pp.
Lilley, J.H. and Roberts, R.J. (1997). Pathogenicity and culture studies comparing the Aphanomyces involved in epizootic ulcerative syndrome (EUS) with other similar fungi. Journal of Fish Diseases 20, 135-144.
Lilley, J.H., Hart, D., Richards, R.H., Roberts, R.J., Cerenius L. and Söderhäll, K. (1997). Pan-Asian spread of single fungal clone results in large scale fish kills. Veterinary Record 140, 653-654.
Malvick, D.K., Grau, C.R. and Percich, J.A. (1998). Characterization of Aphanomyces euteiches strains based on pathogenicity tests and random amplified polymorphic DNA analyses. Mycological Research 102, 465-475.
Molina, F.I., Jong, S.-C. and Ma, G. (1995). Molecular characterization and identification of Saprolegnia by restriction analysis of genes coding for ribosomal RNA. Antonie van Leeuwenhoek 68, 65-74.
Roberts, R.J., Campbell, B. and MacRae, I.H. (1994). Proceedings of the Regional Seminar on Epizootic Ulcerative Syndrome. The Aquatic Animal Health Research Institute, Bangkok, Thailand, 25-27 January 1994.
Willoughby, L.G. and Lilley, J. (1992). The ecology of aquatic fungi in Thailand, and the fish disease relationship. The AAHRI Newsletter 1, 5-6. Aquatic Animal Health Research Institute, Bangkok.
Willoughby, L.G., Roberts, R.J. and Chinabut, S. (1995). Aphanomyces invaderis sp. nov., the fungal pathogen of freshwater tropical fishes affected by epizootic ulcerative syndrome (EUS). Journal of Fish Diseases 18, 273-275.
Yeh, Y. (1989). Inter- and intraspecific restriction fragment length polymorphisms in the genus Aphanomyces. PhD thesis, Texas AandM University.
|
[4] Institute of Aquaculture,
University of Stirling, FK(4LA, Scotland, UK, and [5] Aquatic Animal Health Research Institute, Bangkok, Thailand. |