7.1. General introduction
7.2. Biomass pyrolysis system
7.3. Products and their characteristics
7.4. Pretreatment and characterization of feedstocks
7.5. Pilot rotating cone reactor established in SAU
7.1.1 What is Pyrolysis?
Pyrolysis is thermal degradation either in the complete absence of oxidizing agent, or with such a limited supply that gasification does not occur to an appreciable extent or may be described as partial gasification. Relatively low temperature are employed of 500 to 800 °C, compared to 800 to 1000 °C in gasification. Three products are usually produced: gas, pyrolysis oil and charcoal, the relative proportions of which depend very much on the pyrolysis method, the characteristics of the biomass and the reaction parameters. Fast or flash pyrolysis is used to maximize either gas or liquid products according to the temperature employed.
7.1.2 History of Biomass Pyrolysis
Why is pyrolysis getting interesting now?
There are several ways to make use of the energy contained in the biomass from old direct burning to gasification, pyrolysis. The selection of the most profitable technique to recover the energy from a particular type biomass is and most important step towards a profitable investment.
Direct Combustion is the old way of using biomass. The biomass is completely transformed into heat, but the efficiency is just about 10 percent. The gasification pushes to the maximum level the cracking of biomass by completely transforming it into a combustible gas before burning it. The charcoal production, the slow pyrolysis of wood at temperature 500 °C is a process that charcoal makers have exploited for thousands years. Charcoal is a smokeless fuel which is still used for heating purposes. It first technological use can be dated back to the iron age when charcoal was used in ore melting to produce iron. Production of wood vapor was usually related to the smocking which is one of the oldest food preservation method, probably applied since the development of cooking with fire. These vapors, which contain nature preservatives like formaldehyde and alcohol, were used as feedstocks. The main attraction is small and very simple plants could be made at a very low investment cost. The disadvantage is rather low energy yield and the air pollution.
The biomass pyrolysis is attractive because solid biomass and wastes which are very difficult and costly to manage. can be readily converted into liquid products. These liquids, as crude bio-oil or slurry of charcoal of water or oil, have advantages in transport, storage, combustion, retrofitting and flexibility in production and marketing. The energy densities are summarized in Table 7.1.
The crude pyrolysis oil is a blank fluid which often named as bio-oil, pyrolysis oil, or just oil. The other main product is a slurry which can be made from waste and charcoal with chemical added to stabilize the suspension. Stable and mobile concentration of up to 60% wt have been reported. Slurries can also be made from the oil and charcoal.
In pilot plant, the gas is usually flared but in a commercial process it would be used to drive the process or use it as a fuel drying or power generation.
In transport, the bulk density is important, and some estimated values are given in Table 7.1 Oil and slurry mixture have a clear advantage over wood chips and straw in transport bulk density and notable in energy density.
For longer distance collection of biomass, this difference may be a decisive factor.
Storage and handling may be important because of seasonal variations in production and demand of some storage will always be required. Apart from the bulk density and the energy consideration, it is important that raw biomass will deteriorate during storage due to biological degradation process. Char, however, is very stable and will not biologically degrade. Another important factor is handling, in which liquids have significant advantages over solids.
Generally liquid products are easier to control in the combustion process and this is important in retrofitting existing equipment. Current oil fired burners can not be fully directly with solid biomass without any modification of the unit, which may not be interested in uncertain fuel markets. However, Bio-oil, char-oil slurry and char-water slurries are likely to be required only relatively minor conversion of the equipment or even none in some case. Powered coal burners can relatively easily accept charcoal as a partial fuel replacement, as long as the violative content is compatible with the burner design.
In power station, the gas turbines can readily fired with bio-oil and slurry fuels although there is needed with the alkali ash in the char content of the slurry. Some modified engines can be used to use the upgraded oil. In some countries. there is a market for charcoal lumps and briquettes for leisure and industrial application.
Tab.7.1 Energy and Density Characteristics
|
Feed |
Bulk density Kg/M3) |
Heating value dry basis (GJ/T) |
Energy density (GJ/M3) |
|
straw |
100 |
20 |
2 |
|
woodchips |
400 |
20 |
8 |
|
pyro-oil |
1200 |
25 |
30 |
|
charcoal |
300 |
30 |
9 |
|
char-water slurry (50/50) |
1000 |
15 |
15 |
|
char-oil slurry (20/80) |
1150 |
23 |
26 |
7.1.3 General Introduction of Biomass Pyrolysis Process
So far, there are many kinds of processes of biomass pyrolysis, such as conventional, flash or fast which depend on reaction parameters. However, the typical pyrolysis process can be described as follows:
The biomass are previously cut to size and dried to obtain a fully control of the process. The biomass is therefore feed to the reactor with just enough air to burn that part of biomass or heat carrier (sand or others) supplying the heat necessary to the process. A system of cyclones and condensers allows to recover the products. Generally speaking, the biomass pyrolysis system deals with many aspects: biomass planting, pre-treatment, pyrolysis process, products utilization and upgrading, cost and economic evaluation. The following will review the latest technologies of biomass pyrolysis in European countries and U.S.A.
7.2.1 Classifications of Pyrolysis
Pyrolysis has been practised for centuries for production of charcoal. This requires relatively slow reaction at very low temperatures to maximize solid yield. More recently, studies into the mechanisms of pyrolysis have suggested ways of substantially changing the proportions of the gas, liquid and solid products by changing the rate of heating, temperature and residence time.
High heating rates, of up to a claimed 1000 °C/s or even 10000 °C/s, at temperature below about 650 °C and with rapid quenching, causes the liquid intermediate products of pyrolysis to condense before further reaction breaks down higher molecular weight species into gaseous products. The high reaction rates also minimize char formation, and under some condition no char is apparently formed. At high maximum temperature, the major products is gas. Pyrolysis at these high heating rates is known as fast, or flash pyrolysis according to the heating rate and residence time, although the distinctions are blurred. Other work has attempted to exploit the a complex degradation mechanisms by carry out pyrolysis in unusual environment. The main pyrolysis variants are listed in Table 7.2 and the characteristics of the main models of pyrolysis are summarized into Table 7.3.
Table 7.2 Pyrolysis Technology Variant
|
Tech. |
Residence time |
Heating rate |
Temperature °C |
Products |
|
carbonation |
days |
very low |
400 |
charcoal |
|
Conventional |
5-30 min |
low |
600 |
oil, gas, char |
|
Fast |
0.5-5s |
very high |
650 |
bio-oil |
|
Flash-liquid |
< 1 s |
high |
< 650 |
bio-oil |
|
Flash-gas |
< 1 s |
high |
< 650 |
chemicals, gas |
|
Ultra |
< 0.5 |
very high |
1000 |
chemicals, gas |
|
Vacuum |
2-30s |
medium |
400 |
bio-oil |
|
Hydro-pyro. |
< 10s |
high |
< 500 |
bio-oil |
|
Methano-pyro. |
< 10s |
high |
> 700 |
chemicals |
Table 7.3 Characteristics of Pyrolysis Technologies
|
|
Flash low T |
Flash high T |
Slow |
Carbonization | |
|
Feedstocks |
|||||
|
|
Feedsize |
small |
small |
moderate |
large |
|
|
Moisture |
v.low |
v. low |
low |
low |
|
|
Parameters |
||||
|
|
Temp °C |
450-600 |
650-900 |
500-600 |
450-600 |
|
|
Pressure, bar |
1 |
0.1- 1 |
1 |
1 |
|
|
Max. input, t/h |
0.05 |
0.02 |
5 |
10 |
|
|
Product |
||||
|
|
Gas, % wt dry |
< 30 |
< 70 |
< 40 |
< 40 |
|
|
MJ/Nm3 |
10-20 |
10-20 |
5-10 |
2-4 |
|
|
Liquid % |
< 80 |
< 20 |
< 30 |
< 20 |
|
|
MJ/Kg |
23 |
23 |
23 |
10-20 |
|
|
Solid % |
< 15 |
< 20 |
< 30 |
< 35 |
|
|
MJ/Kg |
30 |
30 |
30 |
30 |
7.2.2 Current Status of the Technologies
In European, a demonstration plant of 500 kg/h is currently operating in Italy for liquid production. There are plans for small commercial units to be derived from this technology in Italy, Spain and Greece as LEBEN projects. A 250 kg/h pilot plant based on the Waterloo processes has been constructed in Spain. Several plants are in operation at a demonstration level for sewage sludge and refuse/WSW in West Germany at capacities up to 2 t/h, based on slow temperature pyrolysis.
Elsewhere, a number of demonstration plants for flash pyrolysis are operation in North America at a scale of up to 25 kg/h with plans for several commercial developments ranging up to 40 kg/h, including a commercial installation planned for California based on the SERI ablative pyrolysis and sewage sludge pyrolysis in Canada and Australia. Examples of current research and development activities are listed in Table 7.4 Some properties that have been reported are summarized and compared in Table7.5.
A. Fixed bed reactor
Charcoal can be produced with a fixed bed reactor in which the biomass feedstock is partially gasified by air. The company Bio-Alternative SA operated a downdraft fixed bed gassier of 1 m diameter and 3 m height (Bridgwater and Bridgw, 1991). with a biomass throughput of 2000kg/h. Products of this process are gas, viscous tars and charcoal of which the yield is maximized. For fir and beech wood, charcoal yields of 300 weight percent on fed wood basis have been achieved. All products are used as energy carriers.
Table 7.5. Characteristics of Various Pyrolysis Technologies for Bio-oil
|
Technology |
GIT |
Ensyn |
laval |
SERI |
Twente | |
|
Temperature [°C] |
500 |
550 |
480 |
510 |
600 | |
|
Pressure [bar a] |
1.0 |
1.0 |
0.01 |
1.0 |
1.0 | |
|
Flow rate [kg/h] |
50 |
50 |
30 |
30 |
12 | |
|
dp [mm] |
0.5 |
0.2 |
10 |
5 |
0.5 | |
|
t gas [s] |
1.0 |
0.4 |
3 |
1 |
0.5 | |
|
t solid [s] |
1.0 |
0.4 |
100 |
0.5 | ||
|
gas yield [wt%] |
30 |
25 |
14 |
35 |
20 | |
|
tar yield [wt%] |
60 |
65 |
65 |
55 |
70 | |
|
Char yield [wt%] |
10 |
10 |
21 |
10 |
10 | |
|
Tar characteristics (on wet basis) | ||||||
|
Density |
1.23 |
1.21 |
1.23 |
1.20 |
1.20 | |
|
Viscosity [cp] |
10 (60c) |
90 (25c) |
5 (40c) |
90 (30c) |
80 (20c) | |
|
C wt% |
39.5 |
45.5 |
49.9 |
54.4 |
43.2 | |
|
H wt% |
7.5 |
7.0 |
7.0 |
5.7 |
8.2 | |
|
0 wt% |
52.6 |
45.4 |
43.0 |
39.8 |
48.6 | |
|
HHV [MJ/Kg] |
24 |
19.3 |
21 |
15 |
25 | |
|
Water in tar [wt%] |
29 |
16 |
18 |
15 |
25 | |
|
Product yield |
||||||
|
|
% wt liquid |
21 |
59 |
66 |
70 | |
|
|
water |
26 |
26 |
10 |
10 | |
|
|
char |
21 |
15 |
14 |
10 | |
|
|
gas |
32 |
- |
10 |
10 | |
Table 7.4 Comparison of Pyrolysis Process Technologies: ranking according to the desired products
|
Technology |
Organization |
Capacity (kg/h) |
Desired Gas/Tar/char |
T (°C) |
|
|
product |
(Wt%) |
||||
|
Fixed bed |
Bio-Alternative |
2000 |
Char |
55/15/30 |
500-800 |
|
Fluid bed |
THEE |
500 |
Gas |
80/10/10 |
650-1000 |
|
Radiation Furnace |
Univ. Zaragoza |
100 |
Gas |
90/8/2 |
1000-2000 |
|
Conventional |
Alten (KTI+ Itaenergy) |
500 |
Tar |
||
|
Circulation fluid bed |
Ensyn Engineering |
30 |
Tar |
25/65/10 |
450-800 |
|
Fast entrained flow |
Georgia Tech Research Ins. |
50 |
Tar |
30/60/10 |
400-550 |
|
Vacuum |
Laval University |
30 |
Tar |
15/65/20 |
250-450 |
|
Vortex reactor |
Solar Energy research Ins. |
30 |
Tar |
35/55/10 |
475-725 |
|
low temperature |
Tubingen University |
10 |
|||
|
Flash fluid bed |
Waterloo University |
3 |
Tar |
20/70/10 |
425-625 |
|
Rotation cone reactor |
Univ. Twente |
10 |
Tar |
20/70/10 |
500-700 |
B. Fluid bed reactor
The well known fluid bed reactor technology has been applied by Kosstrin (1980), Gourtay et al (1987) and Scott et al (1988). Tar yields, produced by a medium scale (100kg/h) fluid bed reactor, are quite low due to cracking of the vapors in the large volumes of bed and freeboard. Fluid bed reactor technology offers good possibilities in gasifying biomass feedstocks with minimum tar formation. In that case, bed material should be selected on basis of optimum catalytic tar cracking behavior. If, however, tar is the product aimed at, a non-catalytic shallow fluid bed should be applied followed by immediate quenching of the gaseous products.
C. Specific technologies for bio-oil production.
Bio-oil production is maximal at medium process temperatures (450-650) and short vapor residence times in the reactor. Useful criteria for selecting pyrolysis technologies for bio-oil production are: i) the bio-oil yield per unit of mass of wood which should be as high as possible, ii) the reactor capacity of the process should be as large enough to limit the number of scale-up steps to full plant capacity. Pyrolysis technologies include in the following survey are selected on the basis of these criteria. Accordingly, it was decided to consider only processes with bio-oil yield larger than 50 weight percent on dry wood basis and a plant capacity of more than 10kg/h. A schematic arrangement of four notable technologies is present in Fig.7.1; their specific features are given in table 5 together with those of the "Twente rotation cone process".
a. Entrained flow reactor
Biomass pyrolysis in an entrained flow reactor has been studied by Gorton et al (1990) at the Georgia Institute of Technology, Atlanta, GA, U.S.A. A flow sheet of their process is given in Fig.7.1a. The vertical reactor tube has a length of 6.4m and an internal diameter of 0.15m. Air and propane are introduced stoichiometrically and combustion in the bottom section of their reactor. The produced hot flue gas flows upwards through the tube while passing the biomass fees point. In this way the thermal energy of the combustion gas is used to heat the biomass particles and, if necessary, provide the heat of the pyrolysis reaction. Typical operation condition are ratio of carrier-gas mass flow over the pyrolysis mass flow of about 4, a reactor inlet temperature of 900 °C, an atmospheric reactor pressure and a reactor throughput of 500 kg.h. The disadvantage is that it needs large amount of carrier gas (nitrogen).
b. Circulating fluid bed reactor.
An upflow circulating fluid reactor has been operated by Ensyn in Ottawa, Canada (Graham, 1988). Fig.7.1b show that the biomass particles and pre-heated sand are fed together in the bottom section of the circulating fluid reactor. Unfortunately there is no literature available reporting the dimension and the flow rates of the preheated carrier gas and sand for this process. Typical operation of this reactor are a temperature 600 ° C and a biomass throughput of 100 kg/h. It is claimed 60% bio-oil can be achieved with poplar wood as the feed stocks. The use of the sand as a heat carrier offers the advantage of a compact construction because of the high heat transfer rate from the sand to biomass particles. Another advantage is the short residence time of gas, by which secondary tar cracking is suppressed. When this reactor becomes scales-up, special attention should be pay to the rapid mixing of biomass particles with solid heat carrier. Again the requirement of carrier gas is a disadvantage.
c. Vacuum furnace reactor
The vacuum pyrolysis of aspen polar in a multiple hearth reactor has been studied by Roy et al (1992, 1993) at the University of Laval, Quebec, Canada. Six heated hearths with a diameter of 0.7 m are staked on top of a total height of 2 m as part of the reactor given in Fig.7.1c. Wood is fed into the top compartment of reactor and transported downwards by gravity and by the action of scrapers which at present in each compartment. If the biomedicine is converted completely, the bottom compartment contains only charcoal which can be easily removed from the reactor. The temperature of top hearth is about 200 °C and increases towards the bottom the reactor where it reaches 400 ° C to achieve a maximum bio-oil products. A vacuum pump is used to keep the reactor pressure at a value of 1 KPa. A difficulty in scaling -up the reactor is necessary installing a large capacity vacuum pump which is sensitive to fouling and also it is very expensive.
d. Vortex reactor
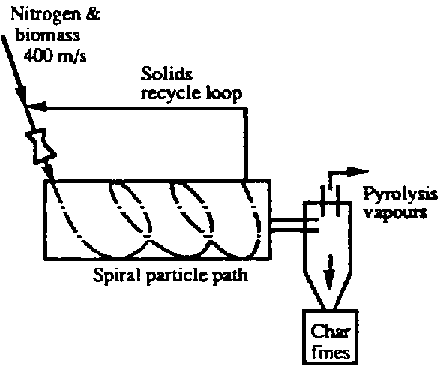
A vortex reactor has been constructed by Diebold and Power (1988) at solar Energy Research Institute, Golden, Co. U.S.A. This reactor has a tube diameter of 0.13 m and a length of 0.7 m For proper operation the reactor, biomass particles should be entrained in a nitrogen flow with velocity of 400 m/s and enter the reactor tube tangentially (see Fig.7.1d). For such condition the biomass particles experience high centrifugal forces which induce high particle ablation rates on the heated reactor wall (625 °C). The ablating particles leave a liquid film of bio-oil on the wall which evaporates rapidly. If the wood particles are not converted completely they may be recycled with a special solids recycle loop. In their paper, Dieblod and Power (1988) estimate the number of cycles required to achieve completely conversion of the biomass particles to be about 15, which is considered to be quit high. However, 80 weight percent bio-oil on dry wood basis has been achieved up to now.
The primary products can be gas, liquid and solid depending on the process employed. Most of the projects interest in the liquid products due to their high energy density and potential for oil substitution.
The liquid, when formed, approximates to biomass in elemental composition with a slight higher heating value of 20-25 MJ/Kg, and is composed of a very complex mixture of oxygenated hydrocarbons. The complexity arise from the degradation of lignin, and the broad spectrum of phenolic compounds. The liquid is often called oil, but is more like tar. This also can be degraded to liquid hydrocarbon fuels. The crude pyrolysis liquid is a thick black tarry fluid with up to 20 % wt water and viscosity as heavy oil.
The solid products from pyrolysis process is char, which has limited application in developed countries for metallurgical and leisure use. An alternative approach to liquid products lies in grinding the car and slurry it with water with a stabilizer. Stable and mobile concentration of up to 60 % wt has been reported. The slurry can also be made from the bio-oil and char, but the maximum solid concentration appears to be 30 %.
The gas product from pyrolysis usually a MHV fuel gas around 15 -22 MJ/NM3. or a LHV fuel gas of around 4-8 MJ/Nm3 from partial gasification depending on feed and processing parameters.
Fig. 7.1 A Schematic Arrangement of Four Notable Technologies. A. Entrained flow reactor (GIT)
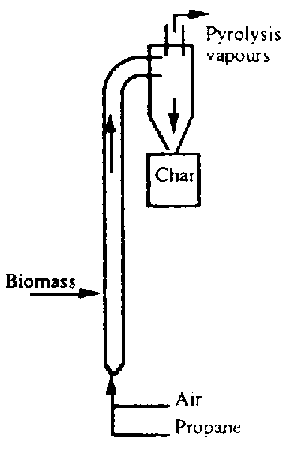
Fig. 7.1 A Schematic Arrangement of Four Notable Technologies. B. Circulating fluid bed reactor (ENSYN)
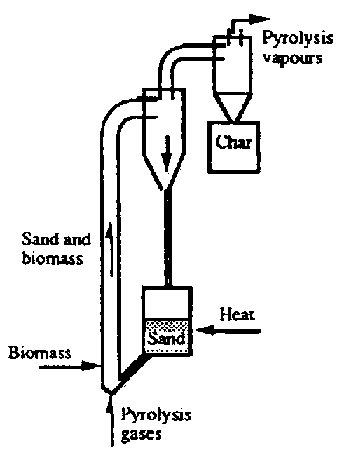
Fig. 7.1 A Schematic Arrangement of Four Notable Technologies. C. Multiple hearth reactor (Univ. Laval)
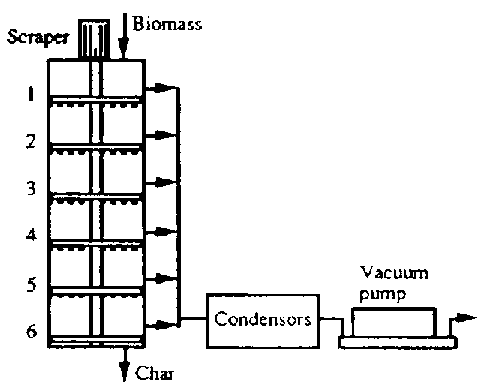
Fig. 7.1 A Schematic Arrangement of Four Notable Technologies. D. Vortex reactor (SERI)
Feedstocks generally considered for thermochemical conversion are wood and wood waste , energy crops such as short rotation forestry and sweet sorghum, agricultural waste and refuse. The main technical criteria for suitability for thermochemical processing are moisture content, ash content and characteristics. The main economic criteria are cost which includes production, collection and transportation, and quantity which includes availability. There is also the question of competing uses such as pulp and board manufacture, combustion, recycle or material recovery rather than energy recovery.
7.4.1 Drying of the Feedstocks
Generally the pyrolysis requires a feedstock at less the 15% moisture content, but there is an optimization to carried out between moisture content and conversion process efficiency. The actual moisture content requires for the conversion process with there for very somewhat between conversion facilities. Biomass as received will have a moisture content typically in the range 50 -60% (wet basis).
Passive drying during summer storage can reduce this to perhaps 30 %. Active silo drying can reduce the moisture content down to 12 %. Drying can be accomplished either by very simple means such as near ambient, solar drying or by waste heat flows or by specifically designed dryers operation the location. Commercial dryers are available in many forms and sites but most common are the rotary kilns and shallow fluidized bed dryers.
7.4.2 Characteristics of Feedstocks
The basic physical characteristics of biomass are summarized in Table 7.6. Particular features the fairly high moisture content, low bulk density and wide particle size range.
Table 7.6 Typical Properties of Feedstocks
|
Feedstocks |
Forestry residues |
process tree |
whole |
MSW |
Straw |
|
moisture content (%) |
30-60 |
20-60 |
40-60 |
15-40 |
10-20 |
|
density (Kg/m3) |
300 |
350 |
300 |
350 |
200 |
7.4.3 Pyrolysis Production Related to the Biomass Composition
The pyrolysis of wood results in the production of gas, tar and char (solid). Of course, the yield of these products are directly related to the composition of the biomass.
Biomass consists of three major components: cellulose, hemicellulose and lignin. Cellulose is a straight and stiff molecule with a polymerization degree of approximately 10.000 glucose unite (C6 sugar) Hemicellulose are polymers built C5, C6 sugars with a polymerisation degree of about 200 sugar units. Both cellulose and hemicellulose can be vapored with negligible char formation at temperatures above 500 "C. Lignin is a three dimensional branched polymer composed phenolic units. Due to the aromatic content of lignin, it degrades slowly on heating and contributes to a major fraction of the char formation. In addition to the major cell wall composition like cellulose, hemicellulose and lignin, biomass often contains varying amounts of species called "extractives". These extractives, which are soluble in polar or no polar solvents, consists of terpenes, fatty acids, aromatic compounds and volatile oil. The composition of various biomass materials is present in Table 7.7
Table 7.7 Composition of Different Biomass Type
|
Type |
CL |
HCL |
LIG |
Extra. |
ASH |
|
Soft wood |
41 |
24 |
28 |
2 |
0.4 |
|
Hard wood |
39 |
35 |
20 |
3 |
0.3 |
|
Pine bark |
34 |
16 |
34 |
14 |
2 |
|
Straw (wheat) |
40 |
28 |
17 |
11 |
7 |
|
Rice husks |
30 |
25 |
12 |
18 |
16 |
|
Peat |
10 |
32 |
44 |
11 |
6 |
note: CL - Cellulose; HCL-Hemicellulose; LIG-lignin
Fig.7.2 shows the processes that govern the biomass particle pyrolysis. First heat is transported to the surface of the particle by conduction. A heated volumetric element within the biomass particle subsequently decomposed into char and vapor fragments which consists of considerable gases (bio-oil) and non- considerable gases. Due to the volumetric vapor production, a pressure is built up inside the porous particle which is at maximum in particle center and decrease towards the particle surface. Vapors which are formed inside the biomass pores are subject to further cracking which leads to the formation of char, gases and thermally stable tars. The long residence time of the vapors inside large particle s at low pyrolysis temperatures explains the formation of charcoal in the case. However this mechanism is absent if the particles size 1 less than 1 mm. When the gaseous products leave the biomass particle, they enter the surrounding gas phase where they can decompose further. Each of these elementary processes is analyzed below in terms of particle properties, process conditions and reactor design.
Fig. 7.2. Sketch of a Decomposing Wood Particle Including the Reaction Paths Involved
The pilot rotating cone reactor of biomass pyrolysis is designed and supplied by Twente University, Netherlands. Its capacity is 50 kg/h. The rotating cone is a new type reactor for flash pyrolysis of biomass to maximize bio-oil production. Wood particles fed to the bottom of the rotating cone together with an excess of inert heat carrier particles, are converted while being transported spirally upwards along the hot cone wall. The geometry of the cone used in the work is specified by a top angle of 90 degree radians and maximum diameter of 650 mm. The most important advantages of the atmospheric rotating cone technology are its high selectivity towards bio-oil and absence of diluting gas. The bio-oil yield is comparable to the yield of other high bio-oil yield production technologies.
The specific features of this reactor are: rapid heating (5000K/s) of the solids, a short residence time of the solids (0.5 s) and a small gas phase residence time (0.3s). Products obtained from the flash pyrolysis of wood dust in a rotation cone reactor are non-condensable gases, bio-oil (tar) and char. Since no carrier gas is needed (cost reducing) the pyrolysis products will be formed at high concentrations. If required. reduction of the gases phase volume inside of the rotating cone is possible by blocking a part of the volume inside the rotating cone; it reduces the gas phase residence time in reactor by which tar cracking in gas phase is suppressed. Figure 7.3 shows a cross section of the reactor in which the rotating cone is visible.
Fig. 7.3 Cross Section of the Rotating Cone Reactor

Conclusions and Problems
The pyrolysis to be the most versatile biomass conversion system, offer high yields of liquid products that can be used directly or upgraded, the technology offers considerably promise for fuels and chemicals, continued R&D is essential to realize the potential.
For the higher liquid product, the more advanced processes are of Twente University, Alten, Waterloo, Tubingen University and Solar Energy Research Institute.
For the integrated system. the following works are still needed to be done:
- Collect data on biomass production, harvesting, conversion and upgrading processes;
- Collect data on transport and handling costs of biomass and derived products
- Continue techno-economic assessment studies for optimization of the system.
- Design the plant towards the lower cost and easy operation.