APPENDIX I. LIST OF PARTICIPANTS - LISTE DES PARTICIPANTS - LISTA DE PARTICIPANTES
APPENDIX II. DRAFT AND DRAFT REVISED MAXIMUM RESIDUE LIMITS FOR PESTICIDES (Advanced to Step 8 of the Codex Procedure)
APPENDIX III. DRAFT REVISED RECOMMENDED METHODS OF SAMPLING FOR THE DETERMINATION OF PESTICIDE RESIDUES FOR COMPLIANCE WITH MRLS (Advanced to Step 8 of the Codex Procedure)
APPENDIX IV. PROPOSED DRAFT AND PROPOSED DRAFT REVISED MAXIMUM RESIDUE LIMITS FOR PESTICIDES (Advanced to Step 5 of the Codex Procedure with Omission of Steps 6 and 7 for Adoption at Step 8)
APPENDIX V. PROPOSED DRAFT AND PROPOSED DRAFT REVISED MAXIMUM RESIDUE LIMITS FOR PESTICIDES AND PROPOSED DRAFT REVISED EXTRANEOUS MAXIMUM RESIDUE LIMIT (Advanced to Step 5 of the Codex Procedure)
APPENDIX VI. CODEX MAXIMUM RESIDUE LIMITS RECOMMENDED FOR REVOCATION
APPENDIX VII. PRIORITY LIST OF COMPOUNDS SCHEDULED FOR EVALUATION OR REEVALUATION BY JMPR
APPENDIX VIII. AGREED CCPR POSITIONS ON ESTIMATION OF EMRLS CCPR RISK MANAGEMENT ISSUES
|
Chairman of the Session: |
Dr W.H. VAN ECK |
|
Président de la Session: |
Ministry of Health, Welfare and Sport |
|
Président de la Reunión: |
PO Box 20350 |
|
|
2500 EJ Den Haag |
|
|
The Netherlands |
|
|
Tel.: + 31 70 3406966 |
|
|
Fax: + 31 70 3405554 |
|
|
e-mail: [email protected] |
Mr. Ouali MOHAMMED-YAHIAOUI
Inspector Général
Ministre Du Commerce
Palais du Gouvernement
Rue du Docteur Cherif Saâdane
Alger 16000
Algeria
tel.: +02 73 74 21
fax: + 02 71 56 91
Abdelkrim HARFOUCHE
Sous-Directeur de la Promotion de la Qualité
Ministre Du Commerce
Palais du Gouvernement
Rue du Docteur Cherif Saâdane
Alger 16000
Algeria
tel.: +02 73 23 40
fax: + 02 71 56 91
Mr Rachid CHENINI
Laboratore du Controle de la Qualité
BP 144 El-Harrach
Gouvernement du Grand Alger
Tel.: +02 52.33.91
Fax: + 02 71 56 91
ARGENTINA
ARGENTINE
Ms Alba R. MUSTACCIOLO
Servicio Nacional de Sanidad y Calidad Agro
Alimentaria
Oficina de Residuos
Prolongacion AvdaBelgrando y Digue II este
1107 Ciudad de Buenos Aires / Argentina
Tel.: (54-1) 312 4015/4050 ext.nr 100 or 188
Fax: (54-1) 312 4015/4050 ext.nr 162
Eduardo BERTI
Embassy of Argentina
Javastraat 20
2585 AN The Hague
Tel.: +31 70 3625907
Fax: +31 70 3469087
Mrs. Maria C. KOCH
Minister
Embassy of Argentina
Javastraat 20
2585 AN THE HAGUE
NETHERLANDS
Tel: +31 7 363 4836
Fax: + 31 7 392 4900
Mr Rubén D. VALLEJO
Counsellor
Embassy of Argentina
Javastraat 20
2585 AN THE HAGUE
NETHERLANDS
Tel: + 31 7 365 4836
Fax:+ 31 7 392 4900
e-mail: [email protected]
AUSTRALIA
AUSTRALIE
Mr. Ian COLEMAN
Chemicals and Biologicals Branch
Food and Agribusiness Industries Division
Edmund Barton Building
GPO Box 858
Canberra ACT 2601
Tel.: +02 6254 9092
Fax: +02 6272 5899
E-mail: [email protected]
Dr. Melanie O’FLYNN
Residue and Standards Brance
National Offices of Food Safety
Department of Agriculture, Fisheries & Forestry
GPO Box 858
CANBERRA ACT 2601
Tel: + 61 2 6272 4549
Fax: + 61 2 6272 4023
E-mail: [email protected]
Ms Nin HYNE
Policy Advisor
Chemicals and Biologicals Branch
Food and Agribusiness Industrie Division
Department of Agriculture, Fisheries & Forestry
GPO Box 858
Canberra ACT 2601
AUSTRALIA
Tel: + 61 2 6272 4301
Fax: + 61 2 6272 5899
e-mail: [email protected]
Dr Angelo A. VALOIS
Section Head Chemical Residues
Policy and International Division
Australian Quarantine and Inspection Service
Department of Agriculture, Fisheries and Forestry
GPO Box 858
Canberra ACT 2601
AUSTRALIA
Tel.: +61 2 6272 5566
Fax: +61 2 6271 6522
e-mail: [email protected]
Ms Janis BAINES
Senior Nutritionist
Australia New Zealand Food Authority
PO Box 7186
Canberra MC ACT 2610
AUSTRALIA
Tel:+ 61 2 6271 2234
Fax: + 61 2 6271 2278
e-mail: [email protected]
Dr Ronald D, EICHNER
Manager Chemical
National Registration Authority
P.O. Box E240
Kingston ACT 2604
Tel.: +61 2 6272 5248
Fax: +61 2 6272 3551
e-mail: [email protected]
Mr. Graham S. ROBERTS
Leader, Organic Chemistry Unit
State Chemistry Laboratory
Department of Natual Resources and Environment
Corner Sneydes and South Roads
Werribee, Victoria 3030
Australia
Tel: + 61 3 9742 8714
Fax: + 61 3 9742 8700
e-mail: [email protected]
Ms Cassie Wright
Old Dept. Primary Industries
Dr. Catherine A. Hollywell
Manager Chemical Standards
Dept of Natural Resources and Environment
5/240 Victoria Parade
East Melbourne, VIC 3002
Tel:+ 61 3 9412 7884
Fax: + 61 3 9412 4775
Dr. Trevor DOUST
Manager Chemistry and Residues Evaluation
National Registration Authority for Agricultural
and Veterinary Chemicals
PO Box E 240
Canberra ACT 2604
Tel.: + 61 2 6272 3208
Fax: + 61 2 6272 3551
E-mail: [email protected]
Dr. Raj BHULA
Agricultural Residues
National Registration Authority
PO Box E240
Kingston ACT 2604
Tel.: +61 2 6272 6551
Fax: +61 2 6272 3551
E-mail: [email protected]
Mr. Bill MURRAY
Grains Research and Development Corporation
22 Thornley Close Ferntree Gully Victoria 3156
Tel.: +61 3 9763 8396
Fax: +61 3 9763 8396
E-mail: [email protected]
Mr Ian F. ECKHARD
Senior Residue Chemist
Agal
PO Box 385
Pymble NSW 2073
Tel: + 61 2 9449 0164
Fax: + 61 2 9449 1653
e-mail: [email protected]
AUSTRIA
AUSTRICHE
Dipl Ing Hermine REICH
Referatsleiterin
Bundesamt und Forschungszentrum für
Landwirtschaft
Institut für Pflanzenschutzmittelprufung
Spargelfeldstraße 191
1226 Wien
Tel.: +43 1/732 16-5130
Fax: +43 1/732 16-5194
E-mail: [email protected]
Mrs. Dr Maria LUSSER
Federal Chancellary
General Directorate VI
Radetzkystrasse 2
1031 VIENNA
Tel: + 43 1 71172 4768
Fax:+ 43 1 71379 52
e-mail: [email protected]
BANGLADESH
Mr.Hossain Delwar
Director
Plant Protection
Department of Agricultural Extension
Khamarbar. Firm-gate, Dhka
BANGLADESH
BELGIUM
BELGIQUE
BELGICA
Ir L. MOHIMONT
Ministère des Classes Moyennes et de
l’Agriculture
Inspection générale Matières
premières et Produits
transformés
WTC 3 - Tour 3
Bd S. Bolivar 30
B-1000 Bruxelles
Tel.: +32 2 208 38 42
Fax: +32 2 208 38 66
Prof dr ir W.P.E. DEJONCKHEERE
Department Crop Protection Chemistry
Faculty Agricultural Science
University Gent
Coupure Links 653
B-9000 Gent
Tel.: +32 9 264 60 09
Fax: +32 9 264 62 47
E-mail: [email protected]
Ir Oliveir N.M.G. PIGEON
Chemist Ingineer / Assistant
Agricultural Research Centre
Phytopharmacy Department
Rue du Bordia 11
B-5030 Gembloux
Tel.: +32 81 625232
Fax: +32 81 62 52 72
E-mail: [email protected]
Dr Christine VINKX
Food Inspector
Ministry of Health
Food Inspection Services
RAC Esplanade, 11th floor
Pachecolaan 19, B5
B-1010 Brussel
Tel.: +32 2 210 48 37
Fax: +32 2 210 48 16
E-mail: [email protected]
BRAZIL
BRESIL
BRASIL
Mr. Luis Antonio SILOS
First Secretary
Embassy of Brazil
Mauritskade 19
2514 HD The Hague
Tel.: +31 70 3023959
Fax: +31 70 3023951
E-mail: [email protected]
Mr. Guilherme Luiz GUIMARAES
Regulatory Specialist
Represent The Brazilian Sindicate of Pesticides
Rua Alexandre Dumas 1671-chac.
Santo Antonio
04717-903
Sao Paulo/SP
BRAZIL
Tel.: +55 11 546 9145
Fax: +55 11 546 9181
E-mail: [email protected]
Mr. Lucas MEDEIROS DANTAS
Technical Consullent
Ministry of Health
Esplanada dos Ministerios, Bloco 6
Ed-Sede 8° Andar
CEP: 70.058-900
Brasilia / DF
Tel.: +55 61 315 2166
Fax: +55 61 315 2727
E-mail: [email protected]
Mr. Alfredo BENATTO
Ministry of Health
National Sanitary Police Agency
Esplanada dos Ministerios
Bloco G. 9° Andar, ED.SEDE
CEP: 70.058.900
Tel.: +5561 315 2619
Fax: +5561 315 2918
E-mail: [email protected]
Mr. Arlindo BONIFÁCIO
Ministry of Agriculture
Esplanada dos Ministerios-Bloco D
Anexo A-3°Andar Sala 350
CEP-70.043-900 Brasilia / DF
Tel: + 55 61 218 2445
Fax: + 55 61 225 5341
E-mail:
[email protected]
Mrs. Heloisa H.B. de TOLEDO
Chemist
Head of Department of Pesticide Residues
Instituto Adolfo Lutz
Av. Dr. Arnaldo 355
01246-902- Sao Paulo - SP
Tel: + 55 11 3064-1527
Fax: + 55 11 3064-1527
E-mail: [email protected]
Mrs. Ivone DELAZARI
Quality Control Manager (Biochemist)
ABIA - Assoc.Bras. da Ind. Alimentos
Av.Brigadeiro Faria Lima, 1478/ll
Sao Paulo / SP
Tel:+ 55 11 816-5733
Fax:+ 55 11 814-6688
E-mail: [email protected]
Mrs. Cleide M.C.M. de OLIVEIRA
Chemist
GARP - Assoc. Grupo de Analistas de Residuo de
Pesticidas
Av. Dr. Arnaldo 355
CEP 01246-902
Sao Paulo / SP
Tel: + 55 11 522-3504
Fax: + 55 11 546-8969
E-mail: [email protected]
Mrs. Rosemarie de S. O. RODRIGUES
Secretary GARP - Assoc. Grupo de Analistas de
Residuo de Pesticidas
Av. Dr Arnaldo 355
Sao Paulo / SP
Tel:+ 55 11 3064-527/ 532-7219
Fax: + 55 11 532-7266
E-mail: [email protected]
Mr Signorin IVENS
Technical Assistant
Mauritskade 19
2514 HD THE HAGUE
Tel: + 31 7 3023959
Fax: + 31 7 3023957
E-mail: [email protected]
CANADA
Mr Bill MURRAY
Health Evaluation Division
Pest Management Regulatory Agency
Health Canada
2250 Riverside Drive Rm D.749
PST Loc. 6607D1
Ottawa, Ontario
K1A OK9
Tel.: +1 61 3 736 3671
Fax: +1 61 3 736 3659
E-mail: [email protected]
Mrs. Louise G. Croteau
Pest Management Regulatory Agency
Health Canada
2250 Riverside Drive Rm E 502
PST Loc. 6605E
Ottawa, Ontario
K1A OK9
Tel.: +1 61 3 736 3536
Fax: +1 61 3 736 3505
Ms Donna GRANT
Chemist, Pesticide Multiresidue Unit
Lab Services - West
Canadian Food Inspection Agency
3650 - 36th Street NW
Calgary, Alberta
T2L 2L1
Tel.: +1 403 299 7636
Fax: +1 403 221 3293
E-mail: [email protected]
CHILE
CHILI
Dr Roberto H. GONZALEZ
Professor of Pesticide Science
University of Chile
College of Agricultural Sciences
P.O. Box 1004
Santiago
Chile
Tel.: +56 2 678 5714
Fax: +56 2 541 7055
E-mail: [email protected]
Sra. Marcela RUIZ
Departamento de Protección Agricola
Servicio Agricola y Ganadero
BULNES 140 3° PISO
Santiago
Chile
Tel.: +56 2 698 22 44 ANEXO 291
Fax: +56 2 696 64 80
E-mail: [email protected]
COSTA RICA
Mrs. Sonia MESÉN JUARÉZ
Jefe del Laboratorio Residuos y Control de Calidad
Ministerio de Agricultura y Ganaderia
Sanidad Vegetal
Apdo 10094
San José
Tel.:+506 260-61-90
Fax:+506.260-83-01
E-mail: [email protected]
CZECH REPUBLIC
REPUBLIQUE TCHEQUE
REPUBLICA CHECA
Mr Helena MALOÑOVÁ
Head of the National Reference Centre of
Pesticides
National Institute of Public Health
Srobárova 48
10000 PRAHA 10
Tel.: +420 2 6708 2377
Fax: +420 2 6731 0291
DENMARK
DANEMARK
DINAMARCA
Mr Arne Buchert
Head of division
Head of delegation
Danish Veterinary and Food Administration
Mørkhøj Bygade 19
DK-2860 Søborg
Tel: +45 339 56461
Fax: +45 339 56696
E-mail: [email protected]
Mr. Milter Green Lauridsen, M.Sc.
Senior Officer
Danish Veterinary and Food Administration
Mørkhøj Bygade 19
DK-2860 Søborg
Tel: +45 339 56464
Fax: +45 339 56696
E-mail: [email protected]
Ms. Hanne Friis Bøtte
Scientific Adviser
Danish Veterinary and Food Administration
Rolighedsvej 25
DK-1958 Frederiksberg C
Tel: +45 339 56204
Fax: +45 339 56680
E-mail: [email protected]
Mr. Per Olsen
Adviser
Danish Agricultural Council
Axeltorv 3
1609 København V
Tel: +45 33 14 56 72
E-mail: [email protected]
EGYPT
EGYPTE
EGIPTO
Dr Sohair Ahmed GAD ALLAH AHMED
Research (Technical Manager) Ministry of
Agriculture
Central Laboratory of Residues Analysis of
Pesticides
and Heavy Metals in Food
14 Nadi El Said St.
Dokki / Giza
Tel.: +20 2 360 1395
Fax: +20 2 361 1216
e-mail: [email protected]
FINLAND
FINLANDE
FINLANDIA
Mr Vesa TUOMAALA
Senior Adviser
Ministry of Trade and Industry
Box 230
00171 Helsinki
Tel.: +358 9 160 3553
Fax: +358 9 160 2648
E-mail: [email protected]
Mr Pekka RAVIO
Chemist
Finnish Customs Laboratory
Tekniikantie 13
SF-02150 Espoo
Tel.: +358 9 614 3276
Fax: +358 9 463 383
E-mail: [email protected]
Ms. Pirjo-Liisa PENTTILA
Senior Scientific Officer
National Food Administration
Box 5
00531 Helsinki
Tel.: +358 9 7726 7621
Fax: +358 9 7726 7666
E-mail: [email protected]
Mr. Hans BLOMQVIST
Head of Division
Plant Production Inspection Center
Pesticide Division
P.O. Box 42
00501 Helsinki
FRANCE
FRANCIA
Mr Jean-Pierre CUGIER
Ministère de l’Agriculture, de la Pêche et
de
l’Alimentation
DGAL/SDPV
INRA/GRAPPA
Domaine Saint Paul
Site Agroparc
84914 AVIGNON CEDEX 9
Tel.: +33 4 9031 6058
Fax: +33 4 9089 6905
Mr Bernard DECLERCQ
Ministère de l’Economie et des Finances
Laboratoire interrégional de la DGCCRF
25, avenue de la République
91305 MASSY CEDEX
Tel.: +33 1 6953 8750
Fax: +33 1 6953 8725
Mrs. Célia BEGUET
Ministère de l’Economie et des Finances
DGCCRF
59, Boulevard Vincent Auriol
75703 Paris Cedex 13
Tel.: + 33 1 44 97 24 68
Fax: +33 1 44 97 30 40
E-mail: [email protected]
Mr. Francois BORDET
Ministère de l’Agriculture et de la
Pêche
CNEVA
43, Rue Danzig
75015 Paris
Tel.: +33 1 49 77 27 37
Fax: +33 1 49 77 26 95
E-mail: [email protected]
Mr Gerard DE CACQUERAY
Agronomist
UIPP - Union des Industries de la Protection des
Plantes
2, rue Denfert-Rochereau
92100 Boulogne-Billancourt
Tel.: +33 1 4002 5321
Fax: +33 1 4345 2819
Mr Michel L’HOTELLIER
Agronomist
UIPP - Union des Industries de la Protection des
Plantes
2, rue Denfert-Rochereau
92100 Boulogne Billancourt
Tel.: +33 1 3081 7381
Fax: +33 1 3081 7251
E-mail: [email protected]
Mr Philippe VERGER
Directeur
Centre de recherches Foch
45, rue des Saint-Pères
75006 Paris
Tel.: +33 1 4296 8421
Fax: +33 1 4020 9685
e-mail: foch@clwb internet.fz
GERMANY
ALLEMAGNE
ALEMANIA
Dr Jutta SCHAUB
Oberregierungsrätin
Bundesministerium für Ernährung,
Landwirtschaft und Forsten
Rochusstrasse 1
D-53123 Bonn
Tel.: 0228 529 3329
Fax: 0228 529 4404
Dr Karsten HOHGARDT
Wissenschaftlicher Oberrat,
Biologische Bundesanstalt für Land- und Forst-
wirtschaft
Messeweg 11/12
D-38104 Braunschweig
Tel.: +49 531 2993503
Fax: +49 531 2993004
E-mail: [email protected]
Dr Renate HANS
Dir.u.Prof., Bundesinstitut für
gesundheitlichen
Verbraucherschutz und Veterinärmedizin
Postfach 33 00 13
D-14191 Berlin
Tel.: +49 30 8412 3383
Fax: +49 30 8412 4741
E-mail:
Dr Lutz ALDER
Wissenschaftlicher Oberrat
Bundesinstitut für gesundheitlichen
Verbraucherschutz und Veterinärmedizin
Postfach 330013
D-141951 Berlin
Tel.: +49 30 8412 0
Fax: +49 30 8412 4741
E-mail: [email protected]
Dr Ursula BANASIAK
Wissenschaftliche Directorin
Biologische Bundesanstalt für Land- und
Forstwirtschaft
Stahnsdorfer Damm 81
D-14532 Kleinmachnow
Tel.: +49 33203 48338
Fax: +49 33203 48425
E-mail: [email protected]
Dr Gabriele TIMME
Bayer AG
Senior Registration Expert
Business Group Crop Protection
Development /Registration
Agrochemical Centre Monheim
D-51368 Leverkussen
Tel.: 00 49 2173 383882
Fax: 00 49 2173 383516
E-mail: [email protected]
Dr. Gudrun OETKEN
Adviser Pesticide Actions Netwerk
Nernstweg 32-34
D-22765 Hamburg
D-22765 Hamburg
Tel.: +49 40 399 19 100
Fax: +49 40 390 7520
E-Mail: [email protected]
Dr Martin SCHAEFFER
Industrieverband Agrar e.v.
Karlstrasse 21
D-60329 Frankfurt/Main
Tel:+ 69 2556 1599
Fax:+ 69 23 6767 02
HUNGARY
HONGRIE
HUNGRIA
Dr Katalin MATYASOVSZKY
Head of the Pesticide Residue Department
National Institute for Food-Hygiene and Nutrition
Gyali ut 3-a
1097 Budapest
Tel.: +361 215 4130
Fax: +361 215 1545
Dr Lászlo GYÖRFI
Deputy Director of Plant Hygiene and
Soil Conservation Station of Budapest
Ministry of Agriculture
Budaörsi ùt 141-145
H-1118 Budapest
Tel.: +36 1 309 1020
Fax: +36 1 1246 2960 / +36 1246 2956
E-mail: [email protected]
INDONESIA
INDONESIE
Dr Kasumbogo UNTUNG
Vice Chairman Pestiside Committee,
Special Assistant
State Ministry of Environment
Jalan D.I. Panjaitan, KEBON NANAS
Jakarta
Tel.: +62 21 858 0107
Fax: +62 21 858 0101
Mr. Sutarto ALIMOESO
Daily Acting Chairman of Pesticides Committee/
Director of Crop Protection
Ministry of Agricultural
Jl. Aup. Pasarminggu
Jakarta Selatan, 72072
Tel:+ 62 21 7806213 / 7819117
Fax:+ 62 1 7805652
E-mail: kompes@ indosat-net.id
Mr. DARYANTO
Secretary of Pesticides Commitee
Ministry of Agricultural
Jl. Aup. Pasarminggu
Jakarta 12520
Tel:+ 62 21 7806213 / 7819117
Fax:+ 62 21 7805652 / 7819117
Mr. Janahar MURAD
Researcher
Ministry of Health Officer
Jl. Percetakan Negara a/o 29
JAKARTA 10560
Tel:+ 62 21 4261088
Fax:+ 62 21 4243933
Mr LAAJURIS
Indonesian Embassy
The Hague
Tel:+ 31 70 3108106
IRELAND
IRLANDE
IRLANDA
Dr Dan O’SULLIVAN
Agricultural Inspector
Pesticide Control Service
Department of Agriculture and Food
Abbotstown
Castleknock
Dublin 15
Tel.: +353 1 607 2614
Fax: +353 1 820 4260
Mr J. QUIGLEY
Senior Chemist
State Laboratory
Abbotstown
Castleknock
Dublin 15
Tel.: +353 1 821 7700
Fax: +353 1 821 7320
ISRAEL
Ms Rina ASHKENAZY
Head of Pesticide Registration Section
Plant Protection and Inspection Services
Ministry of Agriculture
P.O Box 78
Bet-Dagan, 50250
Tel.: +972 3 968 1562
Fax: +972 3 968 1582
E-mail: [email protected]
Dr Rina VARSANO
Head Food Contaminants Section
Ministry of Health
Food Control Administration
P.O. Box 20301
Tel-Aviv 61203
Tel.: +972 3 563 4782 / +972 3 5634837
Fax: +972 3 561 954
E-mail: [email protected]
ITALY
ITALIE
ITALIA
Mr. Ciro IMPAGNATIELLO
Ministero per le Politiche Agricole
VIA XX SETTEMBRE 20
I-00187 ROMA
Italy
Tel.: +39 06 46655016
Fax: +39 06 4880273
JAPAN
JAPON
Ms Mika YOKOTA
Ministry of Agriculture,
Forestry and Fisheries
Kasumigaseki 1-2-1
CHIYODA-KU
TOKYO - 100-8950 Japan
Tel.: +81-3-3501-4094
Fax: + 81-33502-0438
E-mail: [email protected]
Mr. Kazuo OGURA
Senior Inspector
Agricultural Chemicals Inspection Station
2-MM2 Suzuki-Cho
KODAIRA-SHI
TOKYO 187-0011
Japan
Tel.: +81-42-383-2151
Fax: +81-42-385-3361
E-mail: [email protected]
Mr Makoto HIROSE
Deputy Director
Soil and Agricultural Chemicals Division
Waterquality Bureau, Environment Agency
1-2-2, Kasumigaseki
Chiyoda-ku
Tokyo 100-8975
Tel.: +81 3 5521 8321
Fax: +81 3 3593 1438
E-mail: [email protected]
Mr Toshikazu MIYAKAWA
General Manager, International
Japan Grop Protection Association
Nihonbashi Club. 5-8, 1-Chome
Muromachi, Nihonbashi, Chuo-Ku, Tokyo
Tel.: +81 3 3241 0230
Fax: +81 3 3241 3149
E-mail: [email protected]
Ms Kiyomi UENO
Food Sanitation Specialist
Food Chemistry Division
Environmental Health Bureau
Ministry of Health and Welfare
1-2-2, Kasumigaseki,Chiyoda-ku
Tokyo 100-8045
Tel.: +81 3 3595 2341
Fax: +81 3 3501 4868
E-mail:[email protected]
KOREA, REPUBLIC OF
COREE, REPUBLIQUE DE
COREA, REPUBLICA DE
Dr. Chang-Hwan OH
Chief Researcher, Pesticide Division
Food Evaluation Department
Korea Food & Drug Administration
5, Nokbun-Dong, Eunpyung-Ku
Seoul, 122-704
Republic of Korea
Tel.: +82 2 380 1858
Fax: +82 2 382 4892
E-mail: [email protected]
Ms. Jun-Kyung PARK
Researcher of Food Sanitation Council
Korea Food & Drug Administration
5, Nokbun-Dong, Eunpyung-Ku
Seoul, 122-704
Republic of Korea
Tel.: +82 2 380 1564
Fax: +82 2 383 8321
E-mail: [email protected]
Mr. Jae-Seob YOU
Assistant director
Food Policy Division
Ministry of Health and Welfare
1, Chung Ang-Dong, Kwacheon-si
Kyung Ggi-Do
Republic of Korea
Tel.: +82 2 503 7583
Fax: +82 2 503 7534
E-mail: [email protected]
Mr KIM Kyu Yong
Manager
Kyung Nong Corporation 20th.fl
Mijin Plaza B/D 825, Yoksam-Dong
Kangnam-Gu, Seoul 135-080
Republic of Korea
Tel: +82 2 3469 1323
Fax: +82 2 3469 1337
e-mail: [email protected]
Ms SONG Sung-Ok
Veterinary Officer
National Veterinary Research & Quarantine
Service
San 23-4 Deungchon-Dony Kangseo-Gu
Seoul, Republic of Korea
Tel: +82 2 6500 682
Fax:+82 2 6500 655
e-mail: [email protected]
Mr Jae Yeong LEE
Researcher
Kyung Ju Research Institute
Kyung Nong Corporation
226 Kuhwang-Dong, Kyung ju City
Kyung Buk, 780-110
Republic of Korea
Tel: +82 561 776 0133
Fax: +82 561 776 0140
e-mail: [email protected]
Mr Woonhyuck, YOH
Assistant Manager of Dongbu Hannong Chemical
#838, YukSam-Dong, KangNam-Gu,
Seoul, Republic of Korea
Tel: +82 2 3484 1775
Fax: +82 2 568 4702
e-mail: [email protected]
Mr Geon-Jae IM
National Institute of Agricultural Science and
Technology,
Rusal Development Administration
Researcher
249 Seedun-dong Suwoen
Republic of Korea
Tel: +82 331 290 0504
Fax: +82 331 290 521
e-mail: [email protected]
LATVIA
LETTONIE
LETONIA
Mr. Viktors VOLSKIS
National Environment
Centre of Latvia
MIGA Klijánu 7
LV - 1012 Riga
Latvia
Tel.: +3 71 737 0 611
Fax: +3 71 733 9006
E-mail: [email protected]
LESOTHO
Mr. M.T. KHALEMA
Charge D’Affairs of the Lesotho Embassy
Via Serghio 8
00198 Roma
Italy
Tel.: +39 6 8542496
Fax: +39 6 8542527
E-mail: [email protected]
MACEDONIA, THE FORMER YUGOSLAV
REPUBLIC OF
MACEDOINE, L’EX-REPUBLIQUE
YOUGOSLAVE DE
MACEDONIA, LA EX REPUBLICA
YUGOSLAVA DE
Mr. Nenad VEIC
Plant protection Adviser
Ministry of Agriculture, Forestry and Water
Economy
Of the Republic of Macedonia
2 Leninova
91000 Skopje
Republic of Macedonia
Tel.: +389 91 134-477
Fax: + 389 91 230-429
MADAGASCAR
Mr. Hervé Francis RAKOTONDRAVONY
Direction de la Protection des
Végétaux
Service de la Phytopharmacie et du Controle des
Pesticides
Nanisana
Antananarivo 101
Madagascar
Tel.: +261 20 22 402 09
Fax: +261 20 22 41228
E-mail: [email protected]
Mr. Solofo ANDRIATSARAFARA
Responsable des Appuis Scientifiques á
l’Office
National de l’Environnement
BP 822 Antaninarenina 101
Antananarivo, Madagascar
Tel.: +261 20 22 259 99
Fax: +261 20 22 306 93
E-mail: [email protected]
MALAYSIA
MALAISIE
MALASIA
Dr. Cheah Uan BOH
Senior Research Officer
Strategic, Environment & Natural Resources
Research Centre
Malaysian Agricultural Research Development
Institute (MARDI)
PO Box 12301
50774 Kuala Lumpur
Malaysia
Tel: +6 03 9437528
Fax: +6 03 9487639
e-mail: [email protected]
MAURITIUS
MAURICE
MAURICIO
Mr. N. RAMANJOOLOO
Ministry of Agriculture
Agricultural Chemistry Division
Divisional Scientific Officer
Reduit
Mauritius
Tel: +230 465 7472
Fax: +230 464 8749
MEXICO
MEXIQUE
Mrs Amada Velez
Director de Servicior Apoyo Teÿnico
Guillermo Perz Valenzuela # 127
Col - Del Carmen Corjoacon
MEXICO DF
Tel: +525 658 28 28
Fax: +525 658 74 02
e-mail: [email protected]
MOROCCO
MAROC
MARRUECOS
Mr. Lhoussaine SAAD
Ministere de L’Agriculture, du
Développement
Rural et des Pêches Maritimes
DPVCTRF
Station Dbagh°
Avenue Hassan II Rabat
Casablanca
Tel.: +212 7 298150
Fax: +212 7 298150
MYANMAR
Mrs Dr. Khin SAW HLA
Food Control Officer, Food & Drugadministration
35, Min Kyang Street, Dagon Township,
Yangon
Myanmar
Tel: +95 1 245 332
Fax: +95 1 245 331
NETHERLANDS
PAYS-BAS
PAISES BAJOS
Dr ir Henry DE HEER
Senior Official International Phytopharmaceutical
Coordinator
Ministry of Agriculture, Nature Management and
Fisheries
Department of Agriculture
P.O. Box 20401
2500 EK Den Haag
Tel.: +31 70 3785685
Fax: +31 70 3786157
e-mail: [email protected]
Mrs drs P.H. VAN HOEVEN-ARENTZEN
Toxicologist,
National Institute of Public
Health and Environment
P.O. Box 1
3720 BA Bilthoven
Tel.: +31 30 2743263
e-mail: [email protected]
Drs. H. JEURING
Inspectorate for Health Protection
PO Box 16108
2500 BC Den Haag
Tel.: +31 70 3405585
Fax: +31 70 3405435
E-mail:[email protected]
Drs David G. KLOET
Food Safety Adviser
Ministry of Agriculture, Nature Management
and Fisheries
RIKILT-DLO
P.O. Box 230
6700 AE Wageningen
Tel.: +31 317 475 562
Fax: +31 317 417 717
e-mail: [email protected]
Mrs ir Erica MULLER
Consultant Phytopharmacy,
Ministry of Agriculture, Nature
Management and Fisheries
Plant Protection Service
P.O. Box 9102
6700 HC Wageningen
Tel.: +31 317 496 881
Fax: +31 317 421 701
E-mail:[email protected]
Mw. drs. N.M.I. Scheidegger
Ministerie LNV
Directie Veterinaire, Voedings- en
Milieuaangelegenheden
Postbus 20401
2500 EK Den Haag
Tel.: +31 70 3784479
Fax: +31 70 378866141
E-mail: [email protected]
Dr Piet VAN ZOONEN
Head of Laboratory
National Institute of Public Health
and the Environment
P.O. Box 1
3720 BA Bilthoven
Tel.: +31 30 274 2876
Fax: +31 30 274 4424
e-mail: [email protected]
Ir D. van der SCHAAF
ProAgro B.V.
P.O. Box 1180
3600 BB Maarssen
Mrs. Dr. S. BOSMAN-HOEFAKKER
Board of Pesticide Authorization (CTB)
P.O. Box 217
6700 AA Wageningen
Mrs dr Mia A.T. KERKHOFF
P.M. Contaminants
Unilever Research Laboratorium
P.O. Box 114
3130 AC Vlaardingen
Tel.: +31 10 460 5098
Fax: +31 10 460 5671
e-mail: [email protected]
Ir. J. van der Leer
Special Product Safety
Tel.: 010 - 529 5295
Fax: 010 - 522 0903
e-mail: [email protected]
Mrs ir Monique MELLEMA
Commodity Board for Horticulture
P.O. Box 90403
2509 AB Den Haag
Tel.: +31 7 304 1234
Fax: +31 7 347 8181
NEW ZEALAND
NOUVELLE-ZELANDE
NUEVA ZELANDIA
Mr David W. LUNN
National Advisor (Residue Standards)
Ministry of Agriculture and Forestry
P.O.BOX 2526
Wellington
Tel.: +64 4 474 4210
Fax : +64 4 474 4257
E-mail: [email protected]
Dr W.T. (Bill) JOLLY
Counsellor (Veterinary Services)
New Zealand Embassy
37 Observatory Circle
Washington DC 20008
USA
Tel.: +1 202 328 4861
Fax : +1 202 332 4309
E-mail: [email protected]
NIGERIA
Mr.Alhaji Ibrahim SHEHU, mni
Director Federal
Ministry of Commercie & Tourism, P.M.B.
88, Garki, AbuIa. FCT
Nigeria
Tel.: +234 9 234 2771
Mr. Edward OTADAFERUA, IGHO
Assistant Director
Federal Ministry of Commerce & Tourism
Federal Product Inspection Service
Nnewi Building, PMB 1110, APAPA-LAGOS
Tel.: +234 1 5874 914/234 1 545 3282
Mr. M.O. EIGBE
Principal Commercial Officer
Federal Ministry of Commercie & Tourism, P.M.B.
88, Garki, AbuIa, FCT
Nigeria
Tel.: +234 9 882 1510
NORWAY
NORVEGE
NORUEGA
Mr Joralf PAULSEN
Senior executive officer
Food Chemistry and Toxicology Section
Department of Food Law and International Affairs
Norwegian Food Control Authority
P.O.Box 8187.Dep
N-0034 OSLO
Tel.: +47 222 4 6650
Fax : +47 222 4 6699
E-mail: [email protected]
Mr Borge HOLEN
Lab Manager
Plantenforsk
Osloveilen 1
N-1430 As
Tel.: +47 64 97 0390
Fax : +47 64 97 0387
E-mail: [email protected]
PHILIPPINES
FILIPINAS
Dr. Virginia T.D. PACABA
Chief Agriculturist
Bureau of Plant Indsutry, Department of
Agriculture
692 San Andres
Malate, Manila 1004
Tel: +632 524 0708
Fax: +623 523 7154
E-mail: [email protected]
Dr. Susan May F. CALUMPANG
University Researcher
University of the Philippines Los Banos
College of Agriculture
National Crop Protection Center
College Laguna
Philippines 4031
Tel: +63 49 536 0959/2231/0967/0983
Fax: +63 49 536 2409
e-mail: [email protected]
Dr Dalmacio S. SALTING
Agricultural Attache
297 Ave. Moliere
1050 Brussels
Belgium
Tel: +322 340 33 85/344 11 27
Fax: +322 344 1127
e-mail: [email protected]
Dr. Criselda P. PAGLUANAN, D.V.M., Ph D
Chief Laboratory Services Division
National Meat Inspection Commission
Visayas Ave. Diliman
Qeuzon City 1100
Philippines
Tel: +6 432 924 79 77/80
Fax: +6 432 924 31 19
POLAND
POLOGNE
POLONIA
Prof. Jan K. LUDWICKI
Head, Toxicology Department
National Institute of Hygiene
Chocimska str. 24
00-791 Warsaw
Tel.: +48 22 849 70 84
Fax: +48 22 849 74 41
E-mail: [email protected]
Ms M.B. Bozena MARTINEK
Dept. Head of Pesticide Residues Research
Department
Miczurina str. 20
Poznan
Poland
Tel.: +48 61 86 74841
Fax: +48 61 86 76301
PORTUGAL
Enga. Lizete Palavras
Técnica Superior de 1e Classe da
Direcçao-Geral
de Fiscalização e Controlo da Qualidade
Alimentar
Ministerio de Agricultura
Rua Antonio Enes No. 20 1-°
1050 Lisboa
Portugal
Tel.: +351 1 319 2922
Fax: +351 1 319 2919
Enga. Clarisse HENRIQUES
FIPA-Federação das Industrias
Portuguesas Agro-Alimentares
Rua Beato 44
1900 Lisboa
Portugal
Tel.: +351 1 8686171
Fax: +351 1 8688083
E-mail: [email protected]
ROMENIE
ROUMANIE
RUMANIA
Mrs Ana CSUMA
Asociatia de Standardizare Din România
Str. Mendeleev 21-25
70168 Bucure ti 1
ROMÃNIA
Tel: +40 1 211 3296
Fax: +40 1 210 0833
e-mail: [email protected]
Mrs Maria STANCULESCU
Asociatia de Standardizare Din România
Str. Mendeleev 21-25
70168 Bucure ti 1
ROMÃNIA
Tel: +40 1 211 3296
Fax: +40 1 210 0833
e-mail: [email protected]
SLOVAK REPUBLIK
Dr Jana KOVACICOVá, Ph.D.
Head of Quality department
Institute of Preventive and Clinical Medicine
Limbová 14
833 01 Bratislava
tel.: 421 7 4379332
fax: 421 7 54771094
E-mail: [email protected]
SLOVENIA
SLOVENIE
ESLOVENIA
Dr. Marusa ADAMIC
Spec. of hygiene
Head Dept. of Food and Nutrition
Inst. of Public Health of the Rep of Slovenia
CC Point
Trubarjeva 2
1000 Ljubljana
Slovenia
Tel.: +386 061 1323 245
Fax: + 386 061 323 955
E-mail:
SOUTH AFRICA
AFRIQUE DU SUD
SUDAFRICA
Dr J.B. VERMEULEN
Senior Agricultural Management Advisor
Directorate: Agricultural Production Imputs
National Department of Agriculture
Private Bag X343
Pretoria 0001
Tel.: +27 12 319 7303
Fax : +27 12 319 7179
Ms. F.W.J. JANSEN VAN RIJSSEN
Deputy Director Foodcontrol
Dept. of Health
Private Bag X828
Pretoria 0001
Tel.: +27 12 312 0154
Fax : +27 12 3264374
E-mail: [email protected]
SPAIN
ESPAGNE
ESPAÑA
Dr Angel YAGUE MARTINEZ DE TEJADA
Ministerio de Agricultura, Pesca y Alimentacion
Jefe Servicio Residuos Plaguicidas
Velazquez 147
28002 Madrid
Tel.: 34 1 34 78273/5
Fax: 34 1 34 78316
Dr Josefina LOMBARDERO VEGA
Jefa del Departemento de Residuos
de la Subdireccion General de Analisis
Ministerio de Agricultura, Pesca y Alimentacion
Paseo Infanta Isabel 1
28071 Madrid
Tel.: 34 1 34 74978
Fax: 34 1 34 74968
Dr Santiago GUTIERREZ DEL ARROYO
Tecnico Superior de la Subdireccion General
de Higiene de los Alimentos
Ministerio de Sanidad y Consumo
Paseo del Prado 18-20
28014 Madrid
Tel.: +91 596 1996
Fax: +91 596 4487
Dr Enrique CELMA
Technical Manager Zeneca Agro
Costa Brava 13
28034 Madrid
Spain
Tel.: 34 1 7344011
Fax: 34 1 7350180
Ms Josefina LOMBARDERO VEGA
Iefe Departamento Residuos Pesticidas
Laboratorio Arbitral Agroalimentario M.A.P.A
Carretera Coruna Km 10,7
Madrid 28023 - Spain
Tel: +34 91 347 4978
Fax: +34 91 347 4968
SUDAN
SOUDAN
Prof. Khalid EL ABBADI
Consultant (Pesticides) Sudanese Standard and
Metrology
Organisation, Ministry of Foreign Trade, Sudan
P.O. Box 518 Wad Medani
Sudan
Tel: +249 51 45129
Fax: +249 51 45128
SWEDEN
SUEDE
SUECIA
Mr Arne ANDERSSON
Chief Government Inspector
National Food Administration
P.O. Box 622
S-751 26 Uppsala
Tel.: +46 18 175641
Fax: +46 18 693321
E-mail: [email protected]
Mr Bengt-Göran ERICSSON
Toxicologist
National Food Administration
P.O. Box 622
S-751 26 Uppsala
Tel.: +46 18 171458
Fax: +46 18 105848
E-mail: [email protected]
Mrs. Ingegärd BERGMAN
Principal Administrative Officer
National Food Administration
Food Standards Division
P.O. Box 622
S-751 26 Uppsala
Tel.: +46 18 175500
Fax: +46 18 105848
E-mail: [email protected]
Ms Monika SCHERE
Ministry of Agriculture
Senior Administrative officer
SE 103 33 Stockholm
Tel.: +46 8405 1315
Fax: +46 8405 4970
E-mail: [email protected]
SWITZERLAND
SUISSE
SUIZA
Dr Claude WÜTHRICH
Head of Section
Federal Office of Public Health,
Division of Food Control
Schwarzenburgstrasse 165
CH-2003 Bern
Tel.: +41 31 322 95 69
Fax: +41 31 322 95 74
E-mail: [email protected]
Dr. Elisabeth Bosshard, Ph D
Deputy of Head Section Plant Protection Products
Federal Office of Swiss Agriculture
Section Plant Protection Products
Matteuhofstrasse 5
3003 Beru
Switzerland
Tel: +41 31 324 9080
Fax: +41 31 322 2634
Dr Werner KOBEL
Swiss Society of Chemical Industry
c/o Novartis Crop Protection AG
R1058-7.48
Postfach
CH-4002 Basel
Tel.: +41 61 697 6239
Fax: +41 61 697 5334
E-mail: [email protected]
Mrs P. Danièle MAGNOLATO
Regulatory Affairs Manager
Nestec ltd
55 Av Nestlé
CH-1800 VEVEY
Tel.: +41 21 924 44 41
Fax: 41 21 924 45 47
E-mail: [email protected]
Dr. Richard Stadler
Nestec ltd
55 Av Nestlé
CH-1800 VEVEY
Tel.: +41 21 924 44 41
Fax: 41 21 924 45 47
THAILAND
THAILANDE
TAILANDIA
Dr Nuansri TAYAPUTCH
Director
Division of Agricultural Toxic Substances
Department of Agriculture
Bangkok 10900
Tel.: 662 5793 579
Fax: 662 5614 695
e-mail: [email protected]
Mr. Lakchai MEENAKANIT
Plant Protection Specialist
Department of Agriculture Extension
Ministry of Agriculture and Cooperatives
Chatujak
Bangkok 10900
Thailand
Tel: +66 2 579 3837
e-mail: [email protected]
Ms. Amphai SATRUSAJANG
Deputy Director
The Office Agricultural Standards and Inspections
Horticulture Research Institute Building
In Kasetsart University Campus
Jatujak, Bangkok 10900
Thailand
Tel: +66 2 9406 497
Fax: +66 2 9407 339
e-mail: [email protected]
Mr Pisan PONGSAPITCH
Standards Officer
Officer of the National Codex Alimentarius
Committee
Thai Industrial Standards Institute
Rama VI Road Ratchathewi
Bangkok 10400
Thailand
tel.: +662 2023 444
fax: +662 2487 987
e-mail: [email protected]
Mrs. Thanitha JAENGPRAI
Manager, Thai Frozen Foods Association
160/194- 7ITF BLDG. SILOM RD
Bangrak BANGKOK 10500
Thailand
Tel: +662 261 2355 622-4
Fax: +662 261 2355 625
E-mail: [email protected]
Ms. Charuayporn TANTIPIPATPONG
President, Thai Food Processors’ Association
170/22 9th Floor Ocean Tower 1 Building
New-Rachada Pisek Road, Klongtoey
Bangkok 10110
Thailand
Tel: +662 261 2684 To6
Fax: +662 261 2996 To7
Ms. Hansa PANYA
Secretary of Tuna Packers Group
Thai Food Processors’ Association
170/22 9th Floor Ocean Tower 1 BLDG
New-Ratchadapisek Road, Klongtoey, BKK 10110
Thailand
Tel: +66 2 26126 84-6
Fax: +66 2 2612996-7
e-mail: [email protected]
UNITED ARAB EMIRATES
EMIRATES ARABES UNIS
EMIRATOS ARABES UNIDOS
Ing. Rashid Saleh AL-MEHREZI
Director of the Central Laboratory
Ministry of Agriculture and Fisheries
PO Box 16054
Al Ain
Tel.: +971 3 832255
Fax: +971 3 832075
Dr. Mohd. Osman ELOBEID
Director of Food and Environment Centre
Al-Ain Munisipality
Al Ain PO Boxc 1003
UAE
Tel: +971 3 624666/625425
Fax: +97 3 636338
UNITED KINGDOM
ROYAUME-UNI
REINO UNIDO
Mrs. K. HOSKIN
Ministry of Agriculture, Fisheries and Food
Pesticide Safety Directorate
Mallard House
Kings Pool
3 Peasholme Green
York. YO1 2 PX
Tel.: +44 1904 455 759
Fax: +44 1904 455 733
Mrs. C. HARRIS
Ministry of Agriculture, Fisheries and Food
Pesticide Safety Directorate
Mallard House
Kings Pool
3 Peasholme Green
York. YO1 7 PX
UK
Tel.: +44 1904 455 906
Fax: +44 1904 455711
e-mail: [email protected]
Mr A.R.C. HILL
Ministry of Agriculture, Fisheries and Food
Central Science Laboratory,
Sand Hutton
York. YO4 1LZ
Tel.: +44 1904 462 560
Fax: +44 1904 462 111
e-mail: [email protected]
Mr G. TELLING
Food and Drink Federation
E/o Green End Farmhouse
Perten Hall
Beds. MK44 2AX
UK
Tel.: +44 1480 860 439
Fax: +44 1480 861 739
E-mail: [email protected]
Mr J.R. COX
National Resources Institute
Central Avenue
Chatham Maritime
Kent ME4 4TB
Tel.: +44 1634 883 896
Fax: +44 1634 883 232
e-mail: [email protected]
Mr R. ROWE
European Registration Manager
Dow Elanco
Letcombe Regis
Wantage
Oxon OX12 9JT
Tel.: +44 1235 774 734
Fax: +44 1235 774 749
E-mail:
UNITED STATES OF AMERICA
ETATS-UNIS D’AMERIQUE
ESTADOS UNIDOS D’AMERICA
Mr Fred IVES
Health Effects Division (H7509C)
Office of Pesticide Programs
U.S. Environmental Protection Agency
IOI M 4. S.W.
Washington D.C. 20460
Tel.: +1 703 305 6378
Fax.: +1 703 305 5147
e-mail: [email protected]
Dr Richard M. PARRY, Jr
Assistant Administrator
Agricultural Research Service
U.S. Department of Agriculture
Room 358-A, Administration Bldg.
1400 Independence Ave, SW
Washington DC 20250-0302
Tel.: +1 202 720 3973
Fax: +1 202 720 7549
e-mail: [email protected]
Charles W. COOPER
Director, International Activities Staff Center for
Food Safety and Applied Nutrition
Food and Drug Administration
200 C Street, S.W.
Washington, D.C. 20204
Tel.: +1 202 205 5042
Fax: +1 202 401 7739
mail: [email protected]
Dr. Robert L. EPSTEIN
Associate Deputy Administrator
Science and Technology
Agriculture Marketing Service
U.S. Department of Agriculture
P.O. Box 96456,
Washington DC 20250
Tel.: +1 202 720 2158
Fax: +1 202 720 1484
e-mail: [email protected]
Dr. Robert L. EPSTEIN
Science Advisor Office of the Secretary
US Department of Agriculture
Room 200A Whitten Building
14th Street Independence Avenue
Washingron DC 20250
Tel.: +1 202 720 2593 / 7203631
Fax: + 1 202 720 5437
E-mail: [email protected]
Dr. Stephen FUNK
Health Effects Division (H7509C)
Office of Pesticide Programs
U.S. Environmental Protection Agency
401 M. Street, S.W.
Washington D.C. 20460
Tel.: +1 703 305 5430
Fax.: +1 703 305 5147
e-mail: [email protected]
Ms. Ellen Matten
U.S. Codex Office
Food Safety and Inspection Service
US Department of Agriculture
Room 4861 South Building
1400 Independence Ave. S.W.
Washington, DC 20250-3700
Tel.:+1 202 205 7760
Fax: + 1 202 720 3157
e-mail: [email protected]
Dr. Whang PHANG
Health Effects Division (H7509C)
Office of Pesticide Programs
U.S. Environmental Protection Agency
401 M. Street, S.W.
Washington D.C. 20460
Tel.: +1 703 308 2723
Fax.: +1 703 305 5147
e-mail: [email protected]
Dr. Francis SUHRE
Health Effects Division
Office of Pesticide Programs
Environmental Protection Agency
401 Street, SW (7509C)
Washington, DC 20460
Tel.: +1 703 305 6878
Fax: +1 703 305 5147
e-mail: [email protected]
Dr John P. FRAWLEY
President, Health & Environment International
400 W. 9th Street, Suite 401
Wilmington, Delaware 19801
Tel.: +1 302 426 1717
Fax: +1 302 426 1716
e-mail: [email protected]
Dr Hugh W. EWART
Vice President for Scientific Affairs
Northwest Horticultural Society
650 2nd Street
903 Larson Building
Yakima, WA 98907
Tel.: +1 509 453 3193
Fax: +1 509 457 7615
e-mail: [email protected]
Mr. Chuck ORMAN
Acting President
California Citrus Quality Council
3191 Temple Avenue, Suite No. 115
Pomona, CA 91768-3254
Tel.: +1 909 595 4549
Fax: +1 909 595 7102
e-mail: [email protected]
K.S. RAO, DVM, Ph.D.
Global Risk Assessment Leader
Dow AgroSciences
9330 Zionville Road
Indianapolis, IN 46268-1054
Tel: +1 317 337 4974
Fax: +1 317 337 4567
[email protected]
INTERNATIONAL ORGANIZATIONS
ORGANISATIONS INTERNATIONALES
ORGANIZACIONES INTERNACIONALES
AOAC-INTERNATIONAL
Mr A.R.C. HILL
Ministry of Agriculture, Fisheries and Food
Central Science Laboratory,
Sand Hutton
York. YO4 1LZ
Tel.: +44 1904 462 560
Fax: +44 1904 462 111
e-mail: [email protected]
CONSUMERS INTERNATIONAL (CI)
Ms Lisa Y. LEFFERTS
Consumers International
Head Office
24 Highbury Cresent
London, N5 TRX
United Kingdom
Consumers’ Union
5280, Rockfish Valley Highway
Faber, VA 22938-4001
USA
Tel.: +1 804 361 2420
Fax: +1 804 361 2421
E-mail: [email protected]
[email protected]
Dr Ronald LUIJK
Consumentenbond
PO Box 1000
2500 BA ‘s-Gravenhage
The Netherlands
Tel.: +31 70 445 4366
Fax: +31 70 445 4595
e-mail: [email protected]
Ms. Cristina Tirado
Confederacion de Consumidores y Usarios
c/o Dr. Gomez Ulla, 26, esc. 1, 8-A
28028 Madrid
Spain
Tel.: +34 1 356 0413
Fax: +34 1 356 0413
COUNCIL FOR RESPONSIBLE NUTRITION
Dr. W. Matrin STRAUSS
Director, International Regulator Organization
Monsanto Company
Suite 600
600 13th Street NW
Washington DC 20005
Tel.: +202 383 2845
Fax: +202 783 1924
e-mail: [email protected]
Dr. Russel P. SCHNEIDER
Director Regulatory Affairs
600 13th Street NW
#660
Washington DC 20005
Tel.: +202 383 2866
Fax: +202 183 1924/2468
e-mail: [email protected]
Dr. John CARDELLINA
1875 Eye Street
N.W. Suite 400
Washington DC 20006-5409
Tel.: +202 872 1488
Fax: +202 872 9594
EUROPEAN COMMUNITY (EC)
COMMUNAUTE EUROPEENNE
COMUNIDAD EUROPEA
COMMISSION OF THE EUROPEAN COMMUNITY
Dr. Canice Nolan
Principal Administrator
European Commission
Directorate General VI
DGVI/B/2.1
200 Rue de la Loi
B-1049 Brussels
Tel:+ 32 2 29 61633
Fax: +32 2 29 65963
e-mail: [email protected]
Dr. B. Drukker
Europese Commissie
DG-VI/B/II.1
Rue de la Loi 200
Tel: +32 2 2965779
Fax: +32 2 2965963
e-mail: [email protected]
COUNCIL OF THE EUROPEAN UNION
Mrs. Irène SIMANTONI
Principal Administrator
General Secretariat of the Council of the European
Union
175 Rue de la Loi
B-1048 Brussels
Tel.: +322 285 7702
Fax: +322 285 7928
e-mail: [email protected]
GLOBAL CROP PROTECTION
FEDERATION (GCPF)
P. ADRIAN
FMC Europe
Registration Manager
Avenue Louise 480 B9
1050 Brussels
Belgium
Tel.: +32 2 645 9552
Fax : +32 2 640 6286
E-mail:
Dr M. BLISS, jr
Manager, International Registrations
ISK Biosciences Corporation
5970 Heisley Road
Suite 200
Mentor, OHIO 44060
USA
Tel.: +1 440 357 4651
Fax: +1 440 357 4662
E-mail: [email protected]
Mr M. BUYS
Scientific Advisor
Rhône-Poulenc Agro
Box 9163
FG 9263 LYON CEDEX 09
France
Tel.: +33 472 85 2647
Fax : +33 472 85 2942
E-mail: [email protected]
POULENC.COM
Dr Desmond BYRNE
Executive Director, Registration & Regulatory
Affrs.
Tomen Agro Inc.
100 first Street
San Francisco, CA 94105
USA
Tel.: +1 415 536 3465
Fax: +1 415 284 1884
E-mail: [email protected]
Dr R.R. GAUGHAN
Rohm & Haas
Product Regulatory Manager
Rohm and Haas Company
100 Independence Mall West
PHILADELPHIA, PA 19106
USA
Tel.: +1 215 592 3936
Fax : +1 215 592 3414
E-mail: [email protected]
WillIAM GRAHAM
Registration Manager
Monsanto
270-272 AVE De Tervuren
1150 Brussels
Belgium
Tel.: +32 2 776 4533
Fax : +44 1 386 710143
E-mail: [email protected]
Mr Fujio ISHIJIMA
Central Research Laboratories Toxicology Lab.
Hokko Chemical Industry, Co.Ltd.
2165 Toda, Atougi-shi Kanagawa-Ken
243-0023,
Japan
Tel: +81 462 2805881
Fax: +81 462 28 0164
Dr Bruce G. JULIN
Manager
Du Pont-Belgium
BLDG 3
A. Spinoystraat 6
B-2800 Mechelen
Belgium
Tel.: +32 15 441378
Fax: +32 15 441398
E-mail: [email protected]
Dr M. KAETHNER
Head Dietary Safety Assessment
Novartis Crop Protection
R 1058.800
CH-4002 Basel
Switserland
Tel.: +41 61 69 72849
Fax: +41 61 69 74966
E-mail: [email protected]
Dr. Gerhard KEUCK
Documentation
AGREVO
D-65926 Frankfurt/Main
Germany
Tel.: +49 69 305 3785
Fax: +49 69 305 17290
E-mail: [email protected]
O. KLEIN
Registration Expert
Bayer AG
Business Group Crop Protection
Development Registration
Agrochemical Centre Gronheim
D-51368 Leverkusen
Tel: +44-2173-383463
Fax: +44-2173-383516
Mr J.L. KLEINHANS
Tomen France
18, Avenue de l Opèra
75001 Paris
Tel: +33 1 42961456
Fax: +33 1 42975291
e-mail: [email protected]
Mr Shigeru MARUYAMA
Manager
Sumitomo Chemical Co. Ltd.
27-1, Shinkawa 2-chome, chuo-ku
Tokyo, 104-8260
Tel.: +81 3 5543-5692
Fax: +81 3 5543-5695
E-mail:
Ms. N.L.H.M. Moll
European Crop Protection Association
6 Av. E. van Nieuwenhuyse
1160 Brussels
Belgium
Tel.: + 32 2 663 1550
Fax: + 32 2 663 15 60
E-mail: [email protected]
Dr Richard J. NIELSSON
Director, Strategic Regulatory Issues
American Cyanamid Company
P.O. Box 400
Princeton, N.J. 08543-0400
USA
Tel.: +1 609 716 2354
Fax: +1 609 716 2333
E-mail: [email protected]
Mr M. NOKATA
Chief Manager, Regulatory Affairs Group
Nihon Nohyaku Co. Ltd.
10205 Nihonbashi Chuo-Ku
Tokyo 103
Tel: +3-3274-3383
Fax: +3-3281-2443
e-mail: [email protected]
Ms Yuko OKAMOTO
Manager Reg. & Env. Safety
AGR DuPont K.K.
1-8-1, Shimomegro, Meguro
Tokyo, Japan
Tel.: +81 3 5434 6119
Fax: +81 3 5434 6187
E-mail: [email protected]
Dr Janet OLLINGER
Registration Manager
Rohm and Haas
100 Independence Mall West
Philadelphia, PA 19106
USA
Tel.: +1 215 592 3058
Fax: +1 215 592 3414
E-mail: [email protected]
Mr David J. OSBORN
Registration Specialist
Uniroyal Chemical Limited
Kennet House
4 Langley Quay
Slough
Berkshire SL3 6GEH
Tel.: +44 1753 603056
Fax : +44 1753 603077
E-mail: [email protected]
Mr Frederick John RAVENEY
Director
Agrilex (UK) LTD
P.O. BOX 31
Robertsbridge
E Sussex TN32 5AZ
Tel.: +44 1580 882059
Fax: +44 1580 8825057
E-mail: [email protected]
Dr Falk R. RITTIG
Manager, Int.l Affairs
BASF
P.O. BOX 120
D-67114 Limburgerhof, Germany
Tel.: +49 621 60 27377
Fax: +49 621 60 27701
E-mail: [email protected]
Mr Hirotaka SAKAKIBARA
Rhone-Poulenc Yuka Agro
Rappongi First Bild. 15F
1-9-9 Roppongi Minato-Ku
Tokyo
Tel.: +81 3 5570 6064
Fax : +81 3 5570 6070
E-mail:
Mr M. SAKAKIBARA
Manager
SDS Biotech
Development Department
2-5-6, Shiba, Minato-Ku
Tokyo 105-0014
Tel: +81 3 5427 2417
Fax: +81 3 5427 2432
E-mail: [email protected]
Mr. Toshio SHIMOMURA
ZEN-NOH Agricultural R&D Center
5-5-1 Higashi-Yahata
Hiratsuka Kanagwa
Japan
Tel.: +81 463 22 7701
Fax: +81 463 22 7502
E-mail: [email protected]
Mr Shigeji SUGIMOTO
Assistant Director, Regulatory Affairs
Nippon Soda Co., Ltd.
2-1, 2-Chome, Ohtemachi
Chiyoda-Ku,
100-8165 Tokyo
Tel.: +81 3 3245 6285
Fax : +81 3 3245 6289
E-mail: [email protected]
Mr Yukiharu TANAKA
Manager, Registration & Regulatory Affairs Group
Tomen Corporations.
14-27, Akasaka 2 Chome, Minato-Ku,
Tokyo 107-8677
Tel.: +81 3 3588 7481
Fax: +81 3 3588 9930
E-mail: [email protected]
Mr. John S. THORNTON
Director, Regulatory Affairs
Bayer Cooperation
P.O. Box 4913
Kansas City, Missouri
Tel:+816-242-2255
Fax:+816-242-2738
E-mail: John [email protected]
Mr. G.A. WILLIS
Manager, Regulatory Affairs Dept.
Zeneca, Agrochemicals
Fernhurs
Haslemere
Surrey GU27 3JE
Tel: +44-1428-655604
Fax: +44-1428-655947
E-mail: [email protected]
H. YOSHIDA
Product Registration
Nissan Chemical Industries Ltd.
Agricultural Division
Kowa Hitotsubashi Building
7-1, 3-chome, kanda-Nishiki-cho
Chiyoda-Ku, Tokyo
Tel.: +3296-8151
Fax: +3296-8016
E-mail: [email protected]
Mr Georges De Wilde
EU Regulatory Manager
2, Rue Claude Choffe
69370 Saint-Didier-au-Mont-d’Or
Tel: +33 478643250
Fax: +33 478477005
E-mail:
[email protected]
INTERNATIONAL CO-OPERATIVE
ALLIANCE (ICA)
Mr Hiroshi SUZUKI
Japanese Consumers’ Co-operative Union
Technica Information
3-29-8 Shibuya, Shibuyaku
Tokyo, Japan
Tel.: + 81 3 5778 8109
Fax: + 81 3 5778 8008
e-mail: [email protected]
INTERNATIONAL DAIRY FEDERATION
(IDF)
FEDERATION INTERNATIONALE
DE LAITERIE (FIL)
ir L.G.M.Th. TUINSTRA
c/o Square Vergote
B-1030 Brussels
Belgium
Tel.: +32 2 733 9888
Fax: +32 2 733 0413
e-mail: [email protected]
INSTITUTE OF FOOD TECHNOLOGISTS
(IFT)
Dr Chad B. SANDUSKY
Director, Safety and Exposure Assessment
ENVIRON
4350 North Fairfax Drive
Suite 300
Arlington, VA 22203
Tel.: +703 516 2300
Fax.: +703 516 2345
E-mail: [email protected]
INTERNATIONAL FEDERATION OF FRUIT
JUICES PRODUCERS (IFU)
Mr. Robert J. HISLOP
Senior Scientist
C/o Procter and Gamble GmbH
Sulzbacher Strasse 40
Schwalzbach
Germany
Tel.: +49 6196 89 4962
Fax: +49 6198 89 4476
E-mail: [email protected]
INTERNATIONAL TOXICOLOGY
INFORMATION CENTRE (ITIC)
Dr G. VETTORAZZI
Director International Toxicology Information
Centre (ITIC)
Paseo Ramón María de Lilí, 1, 4°-
D
E-20002 San Sebastian
Spain
Tel: +34 943 320 455
Fax: +34 943 320 487
e-mail: [email protected]
Mr. Alexander A. Avery
Director
National Consumer Coalition
Hudson Institute
P.O. Box 202
Churchville, VA 24421
USA
Tel: +540 337 6354
Fax: +540 337 8593
e-mail: [email protected]
Mrs Leila Barraj
Statisticion
Noviger Sciences, Inc.
1730 Rhode Island Ave. NW
Suite 1100
Washington DC 20036
Tel: + 202 293 5374
Fax: + 202 293 5377
e-mail: [email protected]
Dr Barbara J. PETERSEN
President
Novigen Sciences, Inc.
1730 Rhode Island Avenue N.W.,
# 1100
Washington D.C. 20036
USA
Tel.: +1 202 293 5374
Fax: +1 202 293 5377
E-mail: [email protected]
John R. WESSEL
Health & Environment Int’l
Suite 501
10517 Catterskill Court
Columbia, MD 21044
USA
Tel.: +1 301 854 2501
Fax: +1 301 854 2502
e-mail: [email protected]
INTERNATIONAL UNION OF PURE AND
APPLIED CHEMISTRY (IUPAC)
Dr. Kenneth D. Racke
Global Regulatory Leader
Dow AgroSciences
9330 Zionsville Road
Bldg 308-2B
Indianapolis, IN 46268
USA
Phone: 1-317-337-4654
Fax: 1-317-337-3810
Email: [email protected]
Dr. Sue Sun-Wong
Senior Specialist of Taiwan Agricultural
Chemicals and Toxic Research Institute
11 Kung-Ming Road, Wufeng
Taichung Hsien
TAIWAN
Phone: +886-4-330-2101 ext. 401
Fax: +886-4-332-4738
Email: [email protected]
INTERNATIONAL ATOMIC ENERGY
AGNENCY (IAEA)
Dr. Árpád AMBRUS
Joint FAO/IAEA Division of Nuclear Techniques
in Food and Agriculture
IAEA
Wagramer Strasse 5, PO Box 100
A-1400 Vienna
Austria
Tel.: +43 1 260028395
Fax: +43 1 2600 28222
E-mail: [email protected]
OFFICE INTERNATIONAL DE LA VIGNE
ET DU VIN (OIV)
Mr Dr A.P. Dominique TUSSEAU
Scientific secretary-Oenological Commission
Office International de la Vigne et du Vin
c/o CIVC
5 Rue H Martin -BP 135
51204 Epernay Cedex
France
Tel.: +33 3 26 511930
Fax: +33 3 26 511957
e-mail: [email protected]
FOOD AND AGRICULTURE ORGANIZATION
OF THE UNITED NATIONS (FAO)
ORGANISATION DES NATIONS UNIES POUR
L’ALIMENTATION ET L’AGRICUTURE
ORGANIZACION DE LAS NACIONES UNIDAS
PARA LA AGRICULTURE Y LA
ALIMENTACION
Dr Amelia W. TEJADA
FAO Joint Secretary to JMPR
Plant Production and Protection Division
FAO
Viale delle Caracalla
00100 Rome
Italy
Tel: +3906 570 54010
Fax: +39 06 57 056347
e-mail: [email protected]
Dr Gero VAAGT
Senior Officer, Pesticides Management Group
Plant Production an Protection Div.
Viale delle Caracalla
00100 Rome
Italy
Tel: +39 06 5705 5757
Fax: +39 06 57056347
e-mail: gero.vaagt @fao.org
WORLD HEALTH ORGANIZATION (WHO)
ORGANISATION MONDIALE DE LA SANTE
(OMS)
ORGANIZACION MUNDIAL DE LA SALUD
Dr John L. HERRMAN
International Programme on Chemical Safety
World Health Organization
1211 Geneva 27
Switzerland
Tel: +41 22 791 3569
Fax: +41 22 791 4848
Dr Gerald G. MOY
Programme on Food
World Health Organization
1211 Geneva 27
Switzerland
Tel.: +41 22 791 3698
Fax: +41 22 791 4807
e-mail: [email protected]
JOINT FAO/WHO SECRETARIAT
Dr Yukiko YAMADA
Food Standards Officer
Joint FAO/WHO Food Standards Programme
FAO
Viale delle Terme di Caracalla
00100 Rome Italy
Tel.: +39 06 5705 5443
Fax: +39 06 5705 4593
E-mail: [email protected]
Dr. Jeronimas MASKELIUNAS
Food Standards Officer
Joint FAO/WHO Food Standards Programme
FAO
Viale delle Terme di Caracalla
00100 Rome, Italy
Tel.: +39 06 57053967
Fax: + 39 06 57054593
E-mail: [email protected]
NETHERLANDS SECRETARIAT
SECRETARIAT PAYS-BAS
SECRETARIA PAISES-BAJOS
Drs J.W. DORNSEIFFEN
Ministry of Health, Welfare and Sport
Directorate of Public Health
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3406961
Fax: +31 70 3405554
e-mail: [email protected]
Mrs. K. SCHENKEVELD
Ministry of Health, Welfare and Sport
Directorate of Public Health
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3405188
Fax: +31 70 3405177
e-mail: [email protected]
Ms Sue BAKER
Ministry of Health, Welfare and Sport
Directorate of Public Health
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3406883
Fax: +31 70 3405177
e-mail: [email protected]
Ms Anneke CORTENBACH
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3405188
Fax: +31 70 3405554
e-mail: [email protected]
Drs R. HITTENHAUSEN-GELDERBLOM
Ministry of Health, Welfare and Sport
Inspectorate for Health Protection
Hoogte Kadijk 401
1018 BK Amsterdam
The Netherlands
Tel.: +31 20 524 4600
Fax: +31 20 524 4700
e-mail: [email protected]
Drs N.B. LUCAS LUIJCKX
Ministry of Health, Welfare and Sport
Directorate of Public Health
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3406875
Fax: +31 70 3405177
e-mail: [email protected]
Mrs. T.P. POEPON
Ministry of Health, Welfare and Sport
Directorate of Public Health
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3407285
Fax: +31 70 3407303
e-mail: [email protected]
Ir P.D.A. OLTHOF
Ministry of Health, Welfare and Sport
Directorate of Public Health
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3406955
Fax: +31 70 3405554
e-mail: [email protected]
Drs. A. OTTEVANGER
Ministry of Health, Welfare and Sport
Directorate of Public Health
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3406886
Fax: +31 70 3405554
e-mail: [email protected]
Mevr. Mr. Y. GERNER
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3406663
Fax: +31 70 3405554
e-mail: [email protected]
Mr W. BUITENWEG
Ministry of Health, Welfare and Sport
Directorate of Public Health
P.O. Box 20350
2500 EJ The Hague
The Netherlands
Tel.: +31 70 3405122
Fax: +31 70 3407834
e-mail: [email protected]
|
|
Commodity |
MRL
(mg/kg)[37] |
|
||
|
26 |
DICOFOL |
|
|
|
|
|
ML 106 |
Milks |
0.1 |
F |
|
|
|
58 |
PARATHION |
|
|
|
|
|
FP 226 |
Apple |
0.05 |
(*) |
|
|
|
81 |
CHLOROTHALONIL |
|
|
|
|
|
FS 247 |
Peach |
0.2 |
|
(a) |
|
|
105 |
DITHIOCARBAMATES |
|
|
|
|
|
AM 660 |
Almond hulls |
20 |
|
|
|
|
TN 660 |
Almonds |
0.1 |
(*) |
|
|
|
VS 621 |
Asparagus |
0.1 |
|
|
|
|
FI 327 |
Banana |
2 |
|
(a) |
|
|
GC 640 |
Barley |
1 |
|
|
|
|
AS 640 |
Barley straw and fodder, Dry |
25 |
|
|
|
|
VB 41 |
Cabbages, Head |
5 |
|
|
|
|
VR 577 |
Carrot |
1 |
|
(a) |
|
|
VL 510 |
Cos lettuce |
10 |
|
|
|
|
FB 265 |
Cranberry |
5 |
|
|
|
|
VC 424 |
Cucumber |
2 |
|
(a) |
|
|
FB 21 |
Currants, Black, Red, White |
10 |
|
(a) |
|
|
MO 105 |
Edible offal (mammalian) |
0.1 |
|
|
|
|
PE 112 |
Eggs |
0.05 |
(*) |
|
|
|
VA 381 |
Garlic |
0.5 |
|
|
|
|
DH 1100 |
Hops, Dry |
30 |
|
|
|
|
VL 480 |
Kale |
15 |
|
|
|
|
VA 384 |
Leek |
0.5 |
|
|
|
|
VL 482 |
Lettuce, Head |
10 |
|
(a) |
|
|
AS 645 |
Maize fodder |
2 |
|
|
|
|
FC 3 |
Mandarins |
10 |
|
|
|
|
FI 345 |
Mango |
2 |
|
|
|
|
MM 95 |
Meat (from mammals other than marine mammals) |
0.05 |
(*) |
|
|
|
VC 46 |
Melons, except watermelon |
0.5 |
|
(a) |
|
|
ML 106 |
Milks |
0.05 |
(*) |
|
|
|
VA 385 |
Onion, Bulb |
0.5 |
|
|
|
|
FC 4 |
Oranges, Sweet, Sour |
2 |
|
|
|
|
FI 350 |
Papaya |
5 |
|
|
|
|
SO 697 |
Peanut |
0.1 |
(*) |
|
|
|
AL 697 |
Peanut fodder |
5 |
|
|
|
|
VO 445 |
Peppers, Sweet |
1 |
|
|
|
|
FP 9 |
Pome fruits |
5 |
|
(a) |
|
|
VR 589 |
Potato |
0.2 |
|
(a) |
|
|
PM 110 |
Poultry meat |
0.1 |
|
|
|
|
PO 111 |
Poultry, Edible offal of |
0.1 |
|
|
|
|
VC 429 |
Pumpkins |
0.2 |
|
|
|
|
VA 389 |
Spring onion |
10 |
|
|
|
|
VC 431 |
Squash, Summer |
1 |
|
|
|
|
VR 596 |
Sugar beet |
0.5 |
|
|
|
|
AV 596 |
Sugar beet leaves or tops |
20 |
|
|
|
|
VO 447 |
Sweet corn (corn-on-the-cob) |
0.1 |
(*) |
|
|
|
VO 448 |
Tomato |
5 |
|
(a) |
|
|
VC 432 |
Watermelon |
1 |
|
|
|
|
GC 654 |
Wheat |
1 |
|
(a) |
|
|
AS 654 |
Wheat straw and fodder, Dry |
25 |
|
|
|
|
VC 433 |
Winter squash |
0.1 |
|
|
|
|
178 |
BIFENTHRIN |
|
|
|
|
|
MF 812 |
Cattle fat |
0.5 |
|
|
|
|
ML 812 |
Cattle milk |
0.05 |
(*) |
|
|
|
GC 654 |
Wheat |
0.5 |
Po |
|
|
1. Objective
2. Principles
3. Sampling Procedures
4. Criteria for Determining Compliance
Table 1. Minimum number of primary samples to be taken from a lot
Table 2. Number of randomly selected primary samples required for a given probability of finding at least one non-compliant sample in a lot of meat or poultry, for a given incidence of non-compliant residues in the lot
Table 3. Meat and poultry: description of primary samples and minimum size of laboratory samples
Table 4. Plant products: description of primary samples and minimum size of laboratory samples
Table 5. Egg and dairy products: description of primary samples and minimum size of laboratory samples
ANNEX I. Definition of terms
ANNEX II. A Schematic representation of sampling: meat and poultry
ANNEX II. B Schematic representation of sampling: products other than meat and poultry
ANNEX III. Examples
References
The objective of these sampling procedures is to enable a representative sample to be obtained from a lot, for analysis to determine compliance with Codex Maximum Residue Limits (MRLs) for pesticides.
2.1 Codex MRLs are based on Good Agricultural Practice data and foods derived from commodities that comply with the respective Codex MRLs are intended to be toxicologically acceptable.
2.2 A Codex MRL for a plant, egg or dairy product takes into account the maximum level expected to occur in a composite sample, which has been derived from multiple units of the treated product and which is intended to represent the average residue level in a lot. A Codex MRL for meat and poultry takes into account the maximum level expected to occur in the tissues of individual treated animals or birds.
2.3 In consequence, MRLs for meat and poultry apply to a bulk sample derived from a single primary sample, whereas MRLs for plant products, eggs and dairy products apply to a composite bulk sample derived from 1-10 primary samples.
Notes.
(a) The terms used are defined in Annex I and the procedures are shown schematically in Annexs IIA and IIB.3.1 Precautions to be taken(b) ISO recommendations for sampling of grain1, or other commodities shipped in bulk may be adopted, if required.
Contamination and deterioration of samples must be prevented at all stages, because they may affect the analytical results. Each lot to be checked for compliance must be sampled separately.
3.2 Collection of primary samples
The minimum number of primary samples to be taken from a lot is determined from Table 1, or Table 2 in the case of a suspect lot of meat and . Each primary sample should be taken from a randomly chosen position in the lot, as far as practicable. The primary samples must consist of sufficient material to provide the laboratory sample(s) required from the lot.
Note. (a) Sampling devices required for grain, pulses and tea are described in ISO recommendations and those required for dairy products4 are described by the IDF.3.3 Preparation of the bulk sample
3.3.1 Procedure for meat and poultry (Table 3)
Each primary sample is considered to be a separate bulk sample.
3.3.2 Procedure for plant products, eggs or dairy products (Tables 4 and 5)
The primary samples should be combined and mixed well, if practicable, to form the bulk sample.
3.3.3 Alternative procedure where mixing to form the bulk sample is inappropriate or impractical
Where units may be damaged (and thus residues may be affected) by the processes of mixing or sub-division of the bulk sample, or where large units cannot be mixed to produce a more uniform residue distribution, the units should be allocated randomly to replicate laboratory samples at the time of taking the primary samples. In this case, the result to be used should be the mean of valid results obtained from the laboratory samples analyzed.
3.4 Preparation of the laboratory sample
Where the bulk sample is larger than is required for a laboratory sample, it should be divided to provide a representative portion. A sampling device, quartering, or other appropriate size reduction process may be used but units of fresh plant products or whole eggs should not be cut or broken. Where required, replicate laboratory samples should be withdrawn at this stage or they may be prepared as in 3.3.3, above. The minimum sizes required for laboratory samples are given in Tables 3, 4 and 5.
3.5 Sampling record
The sampling officer must record the nature and origin of the lot; the owner, supplier or carrier of it; the date and place of sampling; and any other relevant information. Any departure from the recommended method of sampling must be recorded. A signed copy of the record must accompany each replicate laboratory sample and a copy should be retained by the sampling officer. A copy of the sampling record should be given to the owner of the lot, or a representative of the owner, whether or not they are to be provided with a laboratory sample. If sampling records are produced in computerised form, these should be distributed to the same recipients and a similar verifiable audit trail maintained.
3.6 Packaging and transmission of the laboratory sample
The laboratory sample must be placed in a clean, inert container which provides secure protection from contamination, damage and leakage. The container should be sealed, securely labelled and the sampling record must be attached. Where a bar code is utilised, it is recommended that alphanumeric information is also provided. The sample must be delivered to the laboratory as soon as practicable. Spoilage in transit must be avoided, e.g. fresh samples should be kept cool and frozen samples must remain frozen. Samples of meat and poultry should be frozen prior to despatch, unless transported to the laboratory before spoilage can occur.
3.7 Preparation of the analytical sample
The laboratory sample should be given a unique identifier which, together with the date of receipt and the sample size, should be added to the sample record. The part of the commodity to be analysed, i.e. the analytical sample, should be separated as soon as practicable. Where the residue level must be calculated to include parts which are not analysed[39], the weights of the separated parts must be recorded.
3.8 Preparation and storage of the analytical portion
The analytical sample should be comminuted, if appropriate, and mixed well, to enable representative analytical portions to be withdrawn. The size of the analytical portion should be determined by the analytical method and the efficiency of mixing. The methods for comminution and mixing should be recorded and should not affect the residues present in the analytical sample. Where appropriate, the analytical sample should be processed under special conditions, e.g. at sub-zero temperature, to minimize adverse effects. Where processing could affect residues and where practical alternative procedures are not available, the analytical portion may consist of whole units, or segments removed from whole units. If the analytical portion thus consists of few units or segments, it is unlikely to be representative of the analytical sample and sufficient replicate portions must be analysed, to indicate the uncertainty of the mean value. If analytical portions are to be stored before analysis, the method and length of time of storage should be such that they do not affect the level of residues present. Additional portions must be withdrawn for replicate and confirmatory analyses, as required.
4.1 Analytical results must be derived from one or more laboratory samples taken from the lot and received in a fit state for analysis. The results must be supported by acceptable quality control data (e.g. for instrument calibration and pesticide recovery - refer to Codex Alimentarius, Volume 2, Section 4.2, “Guidelines on good laboratory practice in pesticide residue analysis”). Results should not be corrected for recovery. Where a residue is found to exceed an MRL, its identity should be confirmed and its concentration must be verified by analysis of one or more additional analytical portions derived from the original laboratory sample(s).
4.2 The Codex MRL applies to the bulk sample.
4.3 The lot complies with a Codex MRL where the MRL is not exceeded by the analytical result(s).
4.4 Where results for the bulk sample exceed the MRL, a decision that the lot is non-compliant must take into account: (i) the results obtained from one or more laboratory samples, as applicable; and (ii) the accuracy and precision of analysis, as indicated by the supporting quality control data.
|
|
Minimum number of primary samples to be taken from the
lot |
||
|
(a) Meat and poultry |
|
||
|
a non-suspect lot |
1 |
||
|
a suspect lot |
determined according to Table 2 |
||
|
(b) Other products |
|
||
|
|
(i) Products, packaged or in bulk, which can be assumed to be
well mixed or homogeneous |
1 see note (d) under definition of a lot, Annex 1 |
|
|
|
(ii) Products, packaged or in bulk, which may not be well
mixed or homogeneous |
see note (i), below |
|
|
either: |
|
||
|
|
Weight of lot, kg |
|
|
|
|
<50 |
3 |
|
|
|
50-500 |
5 |
|
|
|
>500 |
10 |
|
|
or |
|
||
|
|
Number of cans, cartons or other containers |
|
|
|
|
in the lot |
|
|
|
|
1-25 |
1 |
|
|
|
26-100 |
5 |
|
|
|
>100 |
10 |
|
|
Incidence of non-compliant residues in the lot |
Minimum number of samples (no) required to detect a
non-compliant residue with a probability of: |
||
|
% |
90% |
95% |
99% |
|
90 |
1 |
- |
2 |
|
80 |
- |
2 |
3 |
|
70 |
2 |
3 |
4 |
|
60 |
3 |
4 |
5 |
|
50 |
4 |
5 |
7 |
|
40 |
5 |
6 |
9 |
|
35 |
6 |
7 |
11 |
|
30 |
7 |
9 |
13 |
|
25 |
9 |
11 |
17 |
|
20 |
11 |
14 |
21 |
|
15 |
15 |
19 |
29 |
|
10 |
22 |
29 |
44 |
|
5 |
45 |
59 |
90 |
|
1 |
231 |
299 |
459 |
|
0.5 |
460 |
598 |
919 |
|
0.1 |
2302 |
2995 |
4603 |
(a) The Table assumes random sampling.(b) Where the number of primary samples indicated in Table 2 is more than about 10% of units in the total lot, the number of primary samples taken may be fewer and should be calculated as follows:
wher
n = minimum number of primary samples to be taken
no = number of primary samples given in Table 2
N = number of units, capable of yielding a primary sample, in the lot.(c) Where a single primary sample is taken, the probability of detecting a non-compliance is similar to the incidence of non-compliant residues.
(d) For exact or alternative probabilities, or for a different incidence of non-compliance, the number of samples to be taken may be calculated from:
1-p = (1-i)n
where p is the probability and i is the incidence of non-compliant residues in the lot (both expressed as fractions, not percentages), and n is the number of samples.
|
|
Commodity classification |
Examples |
Nature of primary sample to be taken |
Minimum size of each laboratory sample |
|
Class B, primary food commodities of animal
origin |
||||
|
1. |
Mammalian meats, type 06, group 030 |
|||
|
|
Note: for enforcement of MRLs for fat soluble pesticides
samples must be taken according to section 2 below. |
|||
|
1.1 |
Large mammals, whole or half carcass, usually 10 kg or
more |
cattle, sheep, pigs, rabbits |
whole or part of diaphragm, supplemented by cervical muscle,
if necessary |
0.5 kg |
|
1.2 |
Small mammals whole carcass |
|
whole carcass or hind quarters |
0.5 kg, after removal of skin and bone |
|
1.3 |
Mammal meat parts, loose fresh/chilled/frozen packaged
or otherwise |
quarters, chops, steaks, shoulders |
whole unit(s), or a portion of a large unit |
0.5 kg, after removal of bone |
|
1.4 |
Mammal meat parts, bulk frozen |
quarters, chops |
either a frozen cross-section of a container
or the whole(or portions) of individual meat parts |
0.5 kg, after removal of bone |
|
2. |
Mammalian fats, including carcass fat, type 06, group
031 |
|||
|
|
Note: samples of fat taken as described in 2.1, 2.2 and 2.3
may be used to determine compliance of the fat, or the whole product, with the
corresponding MRLs. |
|||
|
2.1 |
Large mammals, at slaughter, whole or half carcass
Usually 10 kg or more |
cattle, sheep, pigs |
kidney, abdominal or subcutaneous fat cut from one
animal |
0.5 kg |
|
2.2 |
Small mammals, at slaughter, whole or half carcass
<10 kg |
|
abdominal or subcutaneous fat from one or more
animals |
0.5 kg |
|
2.3 |
Mammal meat parts |
legs, chops, steaks |
either visible fat, trimmed from
unit(s) |
0.5 kg |
|
|
|
|
or whole unit(s) or portions of whole unit(s),
where fat is not trimmable |
2 kg |
|
2.4 |
Mammal bulk fat tissue |
- |
units taken with a sampling device from at least 3
positions |
0.5 kg |
|
Class B, primary food commodities of animal
origin |
||||
|
3. |
Mammalian offals, type 06, group 032 |
|
|
|
|
3.1 |
Mammal liver, fresh/chilled/frozen |
- |
whole liver(s), or part of liver |
0.4 kg |
|
3.2 |
Mammal kidney, fresh/chilled/frozen |
- |
1 or both kidneys from 1 or more animal |
0.2 kg |
|
3.3 |
Mammal heart, fresh/chilled/frozen |
- |
Whole heart(s), or ventricle portion only, if large |
0.4 kg |
|
|
Commodity classification |
Examples |
Nature of primary sample to be taken |
Minimum size of each laboratory sample |
|
3.4 |
Other mammal offal, fresh/chilled/frozen |
intestines, brains |
Part or whole unit from 1 or more animals, or a cross- section
taken from bulk frozen product |
0.5 kg |
|
4. |
Poultry meats, type 07, group 036 |
|||
|
|
Note: for enforcement of MRLs for fat soluble pesticides
samples must be taken according to section 5 below. |
|||
|
4.1 |
Bird, large-sized carcass >2 kg |
turkey, goose, mature chicken |
thighs, legs and other dark meat |
0.5 kg after removal of skin and bone |
|
4.2 |
Birds, medium-sized carcass 500 g-2 kg |
duckling, guinea fowl, young chicken |
thighs, legs or other dark meat from at least 3
birds |
0.5 kg after removal of skin and bone |
|
4.3 |
Birds, small-sized carcass <500 g carcass |
quail, pigeon |
carcasses from at least6 birds |
0.2 kg of muscle tissue |
|
4.4 |
Bird parts fresh/chilled/frozen, retail or wholesale
packaged |
legs, quarters |
packaged units, or individual parts |
0.5 kg (after removal of skin and bone) |
|
Class B, primary food commodities of animal
origin |
||||
|
5. |
Poultry fats, including carcass fat, type 07, group
037 |
|||
|
|
Note: samples of fat taken as described in 5.1 and 5.2 may be
used to determine compliance of the fat, or the whole product, with the
corresponding MRLs |
|||
|
5.1 |
Birds, at slaughter, whole or part-carcass |
chickens, turkeys, |
units of abdominal fat from at least 3 birds |
0.5 kg |
|
5.2 |
Bird meat parts |
legs, breast muscle |
either visible fat, trimmed from
unit(s) |
0.5 kg |
|
|
|
|
or whole unit(s) or portions of whole unit(s),
where fat is not trimmable |
2 kg |
|
5.3 |
Bird fat tissue in bulk |
- |
units taken with a sampling device from at least 3
positions |
0.5 kg |
|
6. |
Poultry offals, type 07, group 038 |
|||
|
6.1 |
Edible bird offal, except goose and duck fat liver and
similar high value products |
|
units from at least 6 birds, or a cross-section from a
container |
0.2 kg |
|
6.2 |
Goose and duck fat liver and similar high value
products |
|
unit from 1 birds or container |
0.05 kg |
|
|
Commodity classification |
Examples |
Nature of primary sample to be taken |
Minimum size of each laboratory sample |
|
Class E, processed foods of animal origin |
||||
|
7. |
Secondary food commodities of animal origin, type 16,
group 080 dried meats |
|
||
|
|
Derived edible products of animal origin, type 17,
group 085 processed animal fats |
|
||
|
|
Manufactured food (single ingredient) of animal origin,
type 18 |
|
||
|
|
Manufactured food (multi-ingredient) of animal origin,
type 19 |
|
||
|
7.1 |
Mammal or bird, comminuted, cooked canned, dried, rendered,
or otherwise processed products, including multi-ingredient
products |
ham, sausage, minced beef, chicken paste |
packaged units, or a representative cross-section from
a container, or units(including juices, if any) taken with a sampling
device |
0.5 kg or 2 kg if fat content
<5% |
|
|
Commodity classification |
Examples |
Nature of primary samples to be taken |
Minimum size of each laboratory sample |
|
Class A, primary food commodities of plant
origin |
||||
|
1. |
All fresh fruits, type 1, groups 001-008 |
|||
|
|
All fresh vegetables, type 2, groups 009-019, except
group 015 (dry pulses) |
|||
|
1.1 |
small sized fresh products units generally < 25
g |
berries, peas, olives |
whole units, or packages, or units taken with a sampling
device |
1 kg |
|
1.2 |
medium sized fresh products units generally 25-250
g |
apples, oranges |
whole units, |
1 kg (at least 10 units) |
|
1.3 |
large sized fresh products units generally > 250
g |
cabbages, cucumbers, grapes(bunches) |
whole units |
2 kg (at least 5 units) |
|
2. |
Pulses, type 2, group 015 |
soya beans |
|
1 kg |
|
|
Cereal grains, type 3, group 020 |
rice, wheat |
|
1 kg |
|
|
Tree nuts, type 4, group 022 |
except coconuts |
|
1 kg |
|
|
|
coconuts |
|
5 units |
|
|
Oilseeds, type 4, group 023 |
peanuts |
|
500 g |
|
|
Seeds for beverages and sweets, type 4, group
024 |
coffee beans |
|
500 g |
|
3. |
Herbs, type 5, group 027(for dried herbs see: Class
D, type 12, in section 5 of this Table) |
fresh parsley |
whole units |
0.5 kg |
|
|
|
others, fresh |
|
0.2 kg |
|
|
Spices, type 5, group 028 |
dried |
whole units or taken with a sampling device |
0.1 kg |
|
Class C, primary animal feed commodities |
||||
|
4. |
Primary feed commodities of plant origin, type
11 |
|||
|
4.1 |
Legume animal feeds, and other forages and
fodders |
|
whole units, or units taken with a sampling device |
1 kg (at least 10 units) |
|
4.2 |
Straw, hay and other dried products |
|
units taken with a sampling device |
0.5 kg (at least 10
units) |
|
|
Commodity classification |
Examples |
Nature of primary samples to be taken |
Minimum size of each laboratory sample |
|
Class D, processed foods of plant origin |
||||
|
5. |
Secondary food commodities of plant origin, type 12,
dried fruits, vegetables, herbs, milled cereal products |
|||
|
|
Derived products of plant origin, type 13, teas,
vegetable oils, juices, by-products for animal feed and miscellaneous
products |
|||
|
|
Manufactured foods (single ingredient) of plant origin,
type 14 |
|||
|
|
Manufactured foods (multi-ingredient) of plant origin,
type 15, including products with ingredients of animal origin where the
ingredient(s) of plant origin predominate(s), and group 078, breads |
|||
|
5.1 |
Products of high unit value |
|
packages or units taken with a sampling device |
0.1 kg* |
|
5.2 |
Solid products of low bulk density |
hops, tea |
packaged units, or units taken with a sampling
device |
0.2 kg |
|
5.3 |
Other solid products |
bread, flour, apple pomace, dried fruit |
packages or other whole units, or units taken with a sampling
device |
0.5 kg |
|
5.4 |
Liquid products |
vegetable oils, juices |
packaged units, or units taken with a sampling
device |
0.5 l or 0.5 kg |
|
* A smaller laboratory sample may be taken from a product of
exceptionally high value but the reason for doing so should be noted in the
sampling record. |
||||
|
|
Commodity classification |
Examples |
Nature of primary samples to be taken |
Minimum size of each laboratory sample |
|
Class B, primary food commodities of animal
origin |
||||
|
1. |
Poultry eggs, type 7, group 039 |
|||
|
1.1 |
Eggs, except quail and similar |
|
whole eggs |
12 whole chicken eggs, 6 whole goose or duck
eggs |
|
1.2 |
Eggs, quail and similar |
|
whole eggs |
24 whole eggs |
|
|
|
|
|
|
|
2. |
Milks, type 6, group 033 |
|
whole unit(s), or unit(s) taken with a sampling
device |
0.5 l |
|
Class E, processed foods of animal origin |
||||
|
3. |
Secondary food commodities of animal origin, type 16,
group 082 skimmed milks, evaporated milks and milk powders |
|||
|
|
Derived edible products of animal origin, type 17,
group 086 milkfats, group 087 butters, butteroils, creams, cream powders,
caseins, etc. |
|||
|
|
Manufactured food (single ingredient) of animal origin,
type 18, group 090 |
|||
|
|
Manufactured food (multi-ingredient) of animal origin,
type 19, group 092 (including products with ingredients of plant origin where
the ingredient(s) of animal origin predominates(s)) |
|||
|
3.1 |
Liquid milks, milk powders, evaporated milks and creams,
creams, dairy ice creams, yoghurts |
|
packaged unit(s), or unit(s) taken with a sampling
device |
0.5 l (liquid) or 0.5 kg (solid) |
|
Notes. (i) Evaporated milks and evaporated creams in bulk
must be mixed thoroughly before sampling, scraping adhering material from the
sides and bottom of containers and stirring well. About 2-3 l should be removed
and again stirred well before removing the laboratory sample. |
||||
|
(ii) Milk powders in bulk should be sampled aseptically,
passing a dry borer tube through the powder at an even rate. |
||||
|
(iii) Creams in bulk should be mixed thoroughly with a
plunger before sampling but foaming, whipping and churning must be
avoided. |
||||
|
3.2 |
Butter and butteroils |
butter, whey butter, low fat spreads containing butter fat,
anhydrous butteroil, anhydrous milkfat |
whole or parts of packaged unit(s), or unit(s) taken
with a sampling device |
0.2 kg or 0.2 l |
|
3.3 |
Cheeses, including processed cheeses |
|||
|
|
units 0.3 kg or greater |
|
Whole unit(s) or unit(s) cut with a sampling device |
0.5 kg |
|
|
units < 0.3 kg |
|
whole unit(s), or unit(s) cut with a sampling device |
0.3 kg |
|
Note. Cheeses with a circular base should be sampled by
making two cuts radiating from the centre. Cheeses with a rectangular base
should be sampled by making two cuts parallel to the sides. |
||||
|
|
Commodity classification |
Examples |
Nature of primary samples to be taken |
Minimum size of each laboratory sample |
|
3.4 |
Liquid, frozen or dried egg products |
|
unit(s) taken aseptically with a sampling device |
0.5 kg |
Analytical portion
A representative quantity of material removed from the analytical sample, of proper size for measurement of the residue concentration.
Note. A sampling device may be used to withdraw the analytical portion.
Analytical sample
The material prepared for analysis from the laboratory sample, by separation of the portion of the product to be analysed5,6 and then by mixing, grinding, fine chopping, etc., for the removal of analytical portions with minimal sampling error.
Note. Preparation of the analytical sample must reflect the procedure used in setting Codex MRLs and thus the portion of the product to be analysed may include parts that are not normally consumed.
Bulk sample
For products other than meat and poultry, the combined and well mixed aggregate of the primary samples taken from a lot. For meat and poultry, the primary sample is considered to be equivalent to the bulk sample.
Notes.
(a) The primary samples must contribute sufficient material to enable all laboratory samples to be withdrawn from the bulk sample.Laboratory sample(b) Where separate laboratory samples are prepared during collection of the primary sample(s), the bulk sample is the conceptual sum of the laboratory samples, at the time of taking the samples from the lot.
The sample sent to, or received by, the laboratory. A representative quantity of material removed from the bulk sample.
Notes.
(a) The laboratory sample may be the whole or a part of the bulk sample.Lot(b) Units should not be cut or broken to produce the laboratory sample(s), except where subdivision of units is specified in Table 3.
(c) Replicate laboratory samples may be prepared.
A quantity of a food material delivered at one time and known, or presumed, by the sampling officer to have uniform characteristics such as origin, producer, variety, packer, type of packing, markings, consignor, etc. A suspect lot is one which, for any reason, is suspected to contain an excessive residue. A non-suspect lot is one for which there is no reason to suspect that it may contain an excessive residue.
Notes.
(a) Where a consignment is comprised of lots which can be identified as originating from different growers, etc., each lot should be considered separately.Primary sample(b) A consignment may consist of one or more lots.
(c) Where the size or boundary of each lot in a large consignment is not readily established, each one of a series of wagons, lorries, ship’s bays, etc., may be considered to be a separate lot.
(d) A lot may be mixed by grading or manufacturing processes, for example.
One or more units taken from one position in a lot.
Notes.
(a) The position from which a primary sample is taken in the lot should preferably be chosen randomly but, where this is physically impractical, it should be from a random position in the accessible parts of the lot.Sample(b) The number of units required for a primary sample should be determined by the minimum size and number of laboratory samples required.
(c) For plant, egg and dairy products, where more than one primary sample is taken from a lot, each should contribute an approximately similar proportion to the bulk sample.
(d) Units may be allocated randomly to replicate laboratory samples at the time of collecting the primary sample(s), in cases where the units are of medium or large size and mixing the bulk sample would not make the laboratory sample(s) more representative, or where the units (e.g. eggs, soft fruit) could be damaged by mixing.
(e) Where primary samples are taken at intervals during loading or unloading of a lot, the sampling “position” is a point in time.
(f) Units should not be cut or broken to produce the primary sample(s), except where subdivision of units is specified in Table 3.
One or more units selected from a population of units, or a portion of material selected from a larger quantity of material. For the purposes of these recommendations, a representative sample is intended to be representative of the lot, the bulk sample, the animal, etc., in respect of its pesticide residue content and not necessarily in respect of other attributes.
Sampling
The procedure used to draw and constitute a sample.
Sampling device
(i) A tool such as a scoop, dipper, borer, knife or spear, used to remove a unit from bulk material, from packages (such as drums, large cheeses) or from units of meat or poultry which are too large to be taken as primary samples. (ii) A tool such as a riffle box, used to prepare a laboratory sample from a bulk sample, or to prepare an analytical portion from an analytical sample.
Notes.
(a) Specific sampling devices are described by ISO1,2,3 and IDF4 standards.Sampling officer(b) For materials such as loose straw or leaves, the hand of the sampling officer may be considered to be a sampling device.
A person trained in sampling procedures and, where required, authorised by the appropriate authorities to take samples.
Note. The sampling officer is responsible for all procedures leading to and including preparation, packing and shipping of the laboratory sample(s). The officer must understand that consistent adherence to the specified sampling procedures is necessary, must provide complete documentation for samples, and should collaborate closely with the laboratory.
Sample size
The number of units, or quantity of material, constituting the sample.
Unit
The smallest discrete portion in a lot, which should be withdrawn to form the whole or part of a primary sample.
Note. Units should be identified as follows.
(a) Fresh fruit and vegetables. Each whole fruit, vegetable or natural bunch of them (e.g. grapes) should form a unit, except where these are small. Units of packaged small products may be identified as in (d), below. Where a sampling device may be used without damaging the material, units may be created by this means. Individual eggs, fresh fruit or vegetables must not be cut or broken to produce units.(b) Large animals or parts or organs of them. A portion, or the whole, of a specified part or organ should form a unit. Parts or organs may be cut to form units.
(c) Small animals or parts or organs of them. Each whole animal or complete animal part or organ present may form a unit. Where packaged, units may be identified as in (d), below. Where a sampling device may be used without affecting residues, units may be created by this means.
(d) Packaged materials. The smallest discrete packages should be taken as units. Where the smallest packages are very large, they should be sampled as bulk, as in (e), below. Where the smallest packages are very small, a pack of packages may form the unit.
(e) Bulk materials and large packages (such as drums, cheeses, etc.) which are individually too large to be taken as primary samples. The units are created with a sampling device.
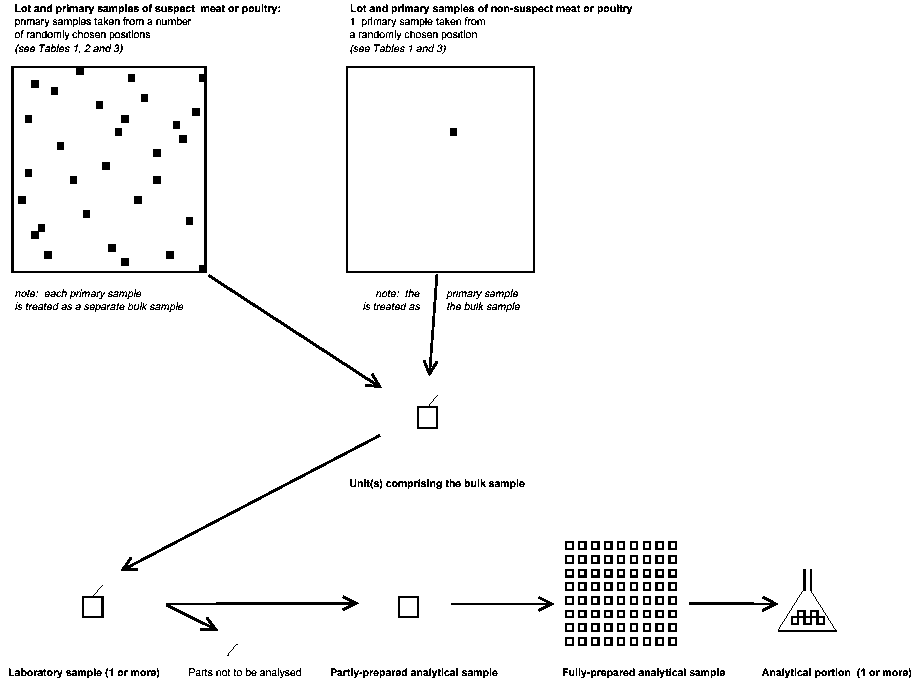
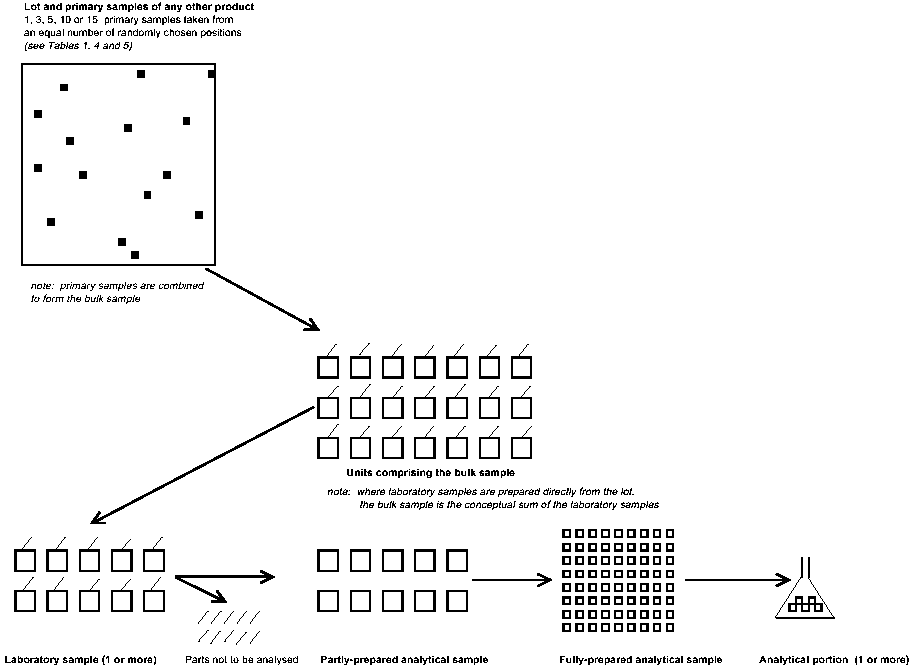
Notes. (i) These examples are provided only as illustrations, they do not form part of the recommendations. (ii) Decisions as to whether or not an MRL is exceeded should be based on the analytical data available but decisions on the consequent action are a matter for the authorities involved.
Example A.
The assumed facts:
1. A 500 t consignment of imported frozen animal carcasses, 300 t labelled as producer A and 200 t labelled as producer B, is to be checked for residues.Consequent actions and decisions:2. The carcasses are from an exporter whose products have recently been associated with excessive residues of permethrin (fat-soluble) and diflubenzuron (non-fat-soluble).
3. Carcasses in lot A have trimmable fat, whereas those in lot B do not.
4. The sampling plan is to provide a 95% probability of detection if 10% of the carcasses contain excessive residues.
5. There is no legal requirement to prepare replicate laboratory samples.
6. Sampling records are in hard copy form.
7. Rendering of fat tissue for extraction of lipid is acceptable under national law.
1. The consignment is sampled as 2 separate, suspect lots, A and B.Example B.2. Table 2 shows that 29 laboratory samples should be taken and therefore, as far as practicable, 29 carcasses are selected at random from each lot.
3. From each selected carcass in lot A, a minimum of 0.5 kg of adhering fat tissue is taken as a (primary) laboratory sample and a minimum of 0.5 kg of meat (meat does not include bone) is taken as a separate (primary) laboratory sample.
4. The carcasses in lot B have no trimmable fat and 29 samples of 2 kg meat are taken.
5. As each laboratory sample is taken, it is placed in a new polythene bag, securely labelled and sealed, and the sample record completed. The samples are sent to the laboratory, ensuring that they do not thaw. Copies of the sample records are given to the owner/custodian of the consignment. Copies are sent with the samples and also retained by the sampling officer.
6. Fat tissue laboratory samples from lot A are rendered, the lipid collected and aliquots (analytical portions) analyzed for permethrin residues. The results are expressed on a whole fat tissue basis.
7. Bones, if any, are removed from the meat laboratory samples, which are minced before the determination of diflubenzuron residues in analytical portions. The results are expressed on the basis of whole meat without bone.
8. If meat samples from both lots contain diflubenzuron £0.05 mg/kg and all samples from lot A contain <1 mg/kg permethrin, lot B is acceptable and lot A is acceptable with respect to diflubenzuron residues.
9. If 3 of the 29 fat samples of lot A contain permethrin >1 mg/kg, replicate analytical portions of fat from these 3 laboratory samples are analyzed. Taking into account the analytical uncertainty, if the results confirm that the MRL is exceeded, the 3 carcasses do not comply with the MRL, whereas the other 26 do comply with the MRL.
10. If the entire lot is not to be rejected on this basis, laboratory samples of fat tissue from the remaining carcasses in lot A may be taken for analysis, in order to separate the acceptable carcasses from those that are unacceptable.
The assumed facts:
1. A consignment of 60 t of apples in 12 kg cartons (each containing approximately 100 apples) is to be checked for residues.Consequent actions and decisions:2. All cartons have the same grower code and date marks.
3. Triplicate laboratory samples are required by national law.
4. The sampling officer is unsure of the degree of mixing that has occurred during packing and grading.
5. Sampling records are in hard copy form.
6. A replicate laboratory sample is held by the monitoring laboratory, until required for analysis by the referee laboratory.
1. The consignment is sampled as a single lot.2. As far as practicable, 10 cartons are selected at random and 3 new polythene bags provided for the laboratory samples.
3. From each carton, apples are taken and placed in each of the bags (1-2 in each), ensuring that in each bag there is a minimum of 10 apples, weighing a total of ³1 kg. The bags are then securely labelled and sealed, and the sample records completed and attached.
4. Two of the laboratory samples are sent to the monitoring laboratory and the third laboratory sample is given to the owner/custodian of the lot.
5. At the monitoring laboratory, the first laboratory sample is prepared and processed and an analytical portion analyzed. The second laboratory sample is retained without further processing.
6. If the results show the confirmed presence of iprodione in excess of the MRL of 10 mg/kg, one or more replicate analytical portion are analyzed.
7. If the results indicate that the MRL is exceeded, the authorities notify the owner/custodian of the consignment (who may arrange independent analysis of the laboratory sample provided) and send the remaining sealed laboratory sample to a reference laboratory.
8. Taking into account the analytical uncertainty at both laboratories, if the results from the reference laboratory indicate residues of iprodione ³10 mg/kg, the MRL is considered to be exceeded.
1. International Organisation for Standardization, 1979. International Standard ISO 950: Cereals - Sampling (as grain).
2. International Organisation for Standardization, 1979. International Standard ISO 951: Pulses in bags - Sampling.
3. International Organisation for Standardization, 1980. International Standard ISO 1839: Sampling - Tea.
4. International Dairy Federation, 1995. International IDF Standard 50C: Milk and milk products - methods of sampling.
5. Joint FAO/WHO Food Standards Programme (1993). “Portion of commodities to which Codex Maximum Residue Limits apply and which is analyzed”. Codex Alimentarius, Volume 2, Section 4.1, 389-404. FAO Rome. ISBN: 92-5-103271-8.
6. Joint FAO/WHO Food Standards Programme (1993). “Codex classification of foods and animal feeds”. Codex Alimentarius, Volume 2, Section 2, 147-366. FAO Rome. ISBN: 92-5-103271-8.
|
|
Commodity |
MRL
(mg/kg)[40] |
|
|
|
|
8 |
CARBARYL[41] |
|
|
|
|
|
AL 1021 |
Alfalfa forage (green) |
100 |
T |
(a) |
|
|
FP 226 |
Apple |
5 |
T |
(a) |
|
|
FS 240 |
Apricot |
10 |
T |
(a) |
|
|
VS 621 |
Asparagus |
10 |
T |
(a) |
|
|
FI 327 |
Banana |
5 |
T |
(a) |
|
|
GC 640 |
Barley |
5 |
Po T |
(a) |
|
|
AL 1030 |
Bean forage (green) |
100 |
T |
(a) |
|
|
VR 574 |
Beetroot |
2 |
T |
(a) |
|
|
FB 264 |
Blackberries |
10 |
T |
(a) |
|
|
FB 20 |
Blueberries |
7 |
T |
(a) |
|
|
VB 41 |
Cabbages, Head |
5 |
T |
(a) |
|
|
VR 577 |
Carrot |
2 |
T |
(a) |
|
|
MM 812 |
Cattle meat |
0.2 |
|
(a) |
|
|
FS 13 |
Cherries |
10 |
T |
(a) |
|
|
FC 1 |
Citrus fruits |
7 |
T |
(a) |
|
|
AL 1023 |
Clover |
100 |
fresh wt T |
(a) |
|
|
VP 526 |
Common bean (pods and/or immature seeds) |
5 |
T |
(a) |
|
|
SO 691 |
Cotton seed |
1 |
T |
(a) |
|
|
VD 527 |
Cowpea (dry) |
1 |
T |
(a) |
|
|
FB 265 |
Cranberry |
7 |
T |
(a) |
|
|
VC 424 |
Cucumber |
3 |
T |
(a) |
|
|
FB 266 |
Dewberries (including boysenberry and loganberry) |
10 |
T |
(a) |
|
|
VO 440 |
Egg plant |
5 |
T |
(a) |
|
|
PE 112 |
Eggs |
0.5 |
T |
(a) |
|
|
MM 814 |
Goat meat |
0.2 |
T |
(a) |
|
|
FB 269 |
Grapes |
5 |
T |
(a) |
|
|
AS 162 |
Hay or fodder (dry) of grasses |
100 |
T |
(a) |
|
|
FI 341 |
Kiwifruit |
10 |
fresh wt T |
(a) |
|
|
VL 53 |
Leafy vegetables |
10 |
T |
(a) |
|
|
AF 645 |
Maize forage |
100 |
T |
(a) |
|
|
VC 46 |
Melons, except watermelon |
3 |
T |
(a) |
|
|
AO3 1 |
Milk products |
0.1 |
(*) T |
(a) |
|
|
ML 106 |
Milks |
0.1 |
(*) T |
(a) |
|
|
FS 245 |
Nectarine |
10 |
T |
(a) |
|
|
AO51900 |
Nuts (whole in shell) |
10 |
T |
(a) |
|
|
GC 647 |
Oats |
5 |
Po T |
(a) |
|
|
VO 442 |
Okra |
10 |
T |
(a) |
|
|
FT 305 |
Olives |
10 |
T |
(a) |
|
|
DM 305 |
Olives, Processed |
1 |
T |
(a) |
|
|
VR 588 |
Parsnip |
2 |
T |
(a) |
|
|
AL 528 |
Pea vines (green) |
100 |
fresh wt T |
(a) |
|
|
FS 247 |
Peach |
10 |
T |
(a) |
|
|
AL 697 |
Peanut fodder |
100 |
T |
(a) |
|
|
SO 703 |
Peanut, Whole |
2 |
T |
(a) |
|
|
AL 697 |
Peanut fodder |
100 |
T |
(a) |
|
|
FP 230 |
Pear |
5 |
T |
(a) |
|
|
VP 63 |
Peas (pods and succulent=immature seeds) |
5 |
T |
(a) |
|
|
VO 51 |
Peppers |
5 |
T |
(a) |
|
|
FS 14 |
Plums (including prunes) |
10 |
T |
(a) |
|
|
VR 589 |
Potato |
0.2 |
T |
(a) |
|
|
PM 110 |
Poultry meat |
0.5 |
V T |
(a) |
|
|
PO 113 |
Poultry skin |
5 |
V T |
(a) |
|
|
VC 429 |
Pumpkins |
3 |
T |
(a) |
|
|
VR 494 |
Radish |
2 |
T |
(a) |
|
|
FB 272 |
Raspberries, Red, Black |
10 |
T |
(a) |
|
|
GC 649 |
Rice |
5 |
PoP T |
(a) |
|
|
CM 649 |
Rice, Husked |
5 |
Po T |
(a) |
|
|
GC 650 |
Rye |
5 |
Po T |
(a) |
|
|
MM 822 |
Sheep meat |
0.2 |
T |
(a) |
|
|
GC 651 |
Sorghum |
10 |
Po T |
(a) |
|
|
AF 651 |
Sorghum forage (green) |
100 |
fresh wt T |
(a) |
|
|
VD 541 |
Soya bean (dry) |
1 |
T |
(a) |
|
|
AL 1265 |
Soya bean forage (green) |
100 |
fresh wt T |
(a) |
|
|
VC 431 |
Squash, Summer |
3 |
T |
(a) |
|
|
FB 275 |
Strawberry |
7 |
T |
(a) |
|
|
VR 596 |
Sugar beet |
0.2 |
T |
(a) |
|
|
AV 596 |
Sugar beet leaves or tops |
100 |
T |
(a) |
|
|
VR 497 |
Swede |
2 |
T |
(a) |
|
|
VO 1275 |
Sweet corn (kernels) |
1 |
T |
(a) |
|
|
VO 448 |
Tomato |
5 |
T |
(a) |
|
|
TN 85 |
Tree nuts |
1 |
T |
(a) |
|
|
GC 654 |
Wheat |
5 |
Po T |
(a) |
|
|
CM 654 |
Wheat bran, Unprocessed |
20 |
PoP T |
(a) |
|
|
CF 1211 |
Wheat flour |
0.2 |
PoP T |
(a) |
|
|
CF 1212 |
Wheat wholemeal |
2 |
PoP T |
(a) |
|
|
VC 433 |
Winter squash |
3 |
T |
(a) |
|
|
65 |
THIABENDAZOLE |
|
|
|
|
|
FI 327 |
Banana |
5 |
Po |
(a) |
|
|
VR 589 |
Potato |
15 |
|
(a) |
|
|
PM 110 |
Poultry meat |
0.05 |
|
|
|
|
VS 469 |
Witloof chicory (sprouts) |
0.05 |
(*) |
|
|
|
81 |
CHLOROTHALONIL |
|
|
|
|
|
VD 71 |
Beans (dry) |
0.2 |
|
|
|
|
HH 624 |
Celery leaves |
3 |
|
|
|
|
FB 21 |
Currants, Black, Red, White |
5 |
|
(a) |
|
|
HH 740 |
Parsley |
3 |
|
|
|
|
VO 445 |
Peppers, Sweet |
7 |
|
|
|
|
VO 447 |
Sweet corn (corn-on-the-cob) |
0.01 |
(*) |
|
|
|
96 |
CARBOFURAN |
|
|
|
|
|
AL 1020 |
Alfalfa fodder |
10 |
|
(a) |
|
|
SB 716 |
Coffee beans |
1 |
|
(a) |
|
|
VR 589 |
Potato |
0.1 |
(*) |
(a) |
|
|
SO 702 |
Sunflower seed |
0.1 |
(*) |
|
|
|
158 |
GLYPHOSATE |
|
|
|
|
|
SO 691 |
Cotton seed |
10 |
|
(a) |
|
|
OC 691 |
Cotton seed oil, Crude |
0.05 |
(*) |
|
|
|
OR 691 |
Cotton seed oil, Edible |
0.05 |
(*) |
|
|
|
GC 645 |
Maize |
1 |
|
(a) |
|
|
AF 645 |
Maize forage |
1 |
|
|
|
|
GC 651 |
Sorghum |
20 |
|
(a) |
|
|
178 |
BIFENTHRIN |
|
|
|
|
|
CM 654 |
Wheat bran, Unprocessed |
2 |
PoP |
|
|
|
CF 1211 |
Wheat flour |
0.2 |
PoP |
|
|
|
CF 1212 |
Wheat wholemeal |
0.5 |
PoP |
|
|
|
181 |
MYCLOBUTANIL |
|
|
|
|
|
FB 278 |
Currant, Black |
0.5 |
|
|
|
|
VO 448 |
Tomato |
0.3 |
|
|
|
|
189 |
TEBUCONAZOLE |
|
|
|
|
|
FI 327 |
Banana |
0.05 |
|
|
|
|
VC 424 |
Cucumber |
0.2 |
|
|
|
|
GC 647 |
Oats |
0.05 |
(*) |
|
|
|
FS 247 |
Peach |
1 |
|
|
|
|
VO 445 |
Peppers, Sweet |
0.5 |
|
|
|
|
FP 9 |
Pome fruits |
0.5 |
|
|
|
|
196 |
TEBUFENOZIDE |
|
|
|
|
|
FI 341 |
Kiwifruit |
0.5 |
|
|
|
|
197 |
FENBUCONAZOLE |
|
|
|
|
|
FI 327 |
Banana |
0.05 |
|
|
|
|
FS 13 |
Cherries |
1 |
|
|
|
|
VC 424 |
Cucumber |
0.2 |
|
|
|
|
FB 269 |
Grapes |
1 |
|
|
|
|
VC 46 |
Melons, except watermelon |
0.2 |
|
|
|
|
TN 672 |
Pecan |
0.05 |
(*) |
|
|
|
FP 9 |
Pome fruits |
0.1 |
|
|
|
|
GC 650 |
Rye |
0.1 |
|
|
|
|
VC 431 |
Squash, Summer |
0.05 |
|
|
|
|
SO 702 |
Sunflower seed |
0.05 |
(*) |
|
|
|
GC 654 |
Wheat |
0.1 |
|
|
|
|
AS 654 |
Wheat straw and fodder, Dry |
3 |
|
|
|
|
|
Commodity |
MRL
(mg/kg)[42] |
|
|
|
|
7 |
CAPTAN |
|
|
|
|
|
FP 226 |
Apple |
20 |
|
|
|
|
AB 226 |
Apple pomace, Dry |
2 |
|
|
|
|
FS 13 |
Cherries |
40 |
|
|
|
|
DF 269 |
Dried grapes (=currants, raisins and sultanas) |
50 |
|
|
|
|
FB 269 |
Grapes |
25 |
|
|
|
|
FS 245 |
Nectarine |
5 |
|
|
|
|
FP 230 |
Pear |
10 |
|
|
|
|
FS 14 |
Plums (including prunes) |
5 |
|
|
|
|
FB 275 |
Strawberry |
30 |
|
|
|
|
VO 448 |
Tomato |
2 |
|
|
|
|
53 |
MEVINPHOS |
|
|
|
|
|
VB 41 |
Cabbages, Head |
0.05 |
|
(a) |
|
|
VP 526 |
Common bean (pods and/or immature seeds) |
0.05 |
|
(a) |
|
|
VA 384 |
Leek |
0.02 |
(*) |
|
|
|
65 |
THIABENDAZOLE |
|
|
|
|
|
MM 812 |
Cattle meat |
0.05 |
|
(a) |
|
|
ML 812 |
Cattle milk |
0.05 |
|
(a) |
|
|
MO 812 |
Cattle, Edible offal of |
0.1 |
|
(a) |
|
|
81 |
CHLOROTHALONIL |
|
|
|
|
|
FI 327 |
Banana |
0.01 |
(*) |
(a) |
|
|
96 |
CARBOFURAN |
|
|
|
|
|
AL 1021 |
Alfalfa forage (green) |
10 |
|
(a) |
|
|
VC 4199 |
Cantaloupe |
0.2 |
|
|
|
|
AB 1 |
Citrus pulp, Dry |
2 |
|
|
|
|
VC 424 |
Cucumber |
0.3 |
|
|
|
|
FC 4 |
Oranges, Sweet, Sour |
0.5 |
|
|
|
|
GC 651 |
Sorghum |
0.1 |
|
|
|
|
AF 651 |
Sorghum forage (green) |
2 |
|
|
|
|
AS 651 |
Sorghum straw and fodder, Dry |
0.5 |
|
|
|
|
VC 431 |
Squash, Summer |
0.3 |
|
|
|
|
VO 447 |
Sweet corn (corn-on-the-cob) |
0.1 |
|
|
|
|
103 |
PHOSMET |
|
|
|
|
|
FS 240 |
Apricot |
10 |
|
(a) |
|
|
SO 691 |
Cotton seed |
0.05 |
|
|
|
|
VR 589 |
Potato |
0.05 |
(*) |
(a) |
|
|
145 |
CARBOSULFAN |
|
|
|
|
|
AB 1 |
Citrus pulp, Dry |
0.1 |
|
|
|
|
FC 4 |
Oranges, Sweet, Sour |
0.1 |
|
|
|
|
177 |
ABAMECTIN |
|
|
|
|
|
AM 660 |
Almond hulls |
0.1 |
|
|
|
|
TN 660 |
Almonds |
0.01 |
(*) |
|
|
|
FP 226 |
Apple |
0.02 |
|
|
|
|
MF 812 |
Cattle fat |
0.1 |
V |
|
|
|
MO 1280 |
Cattle kidney |
0.05 |
V |
|
|
|
MO 1281 |
Cattle liver |
0.1 |
V |
|
|
|
DH 1100 |
Hops, Dry |
0.1 |
|
|
|
|
VL 483 |
Lettuce, Leaf |
0.05 |
|
|
|
|
VC 46 |
Melons, except watermelon |
0.01 |
(*) |
|
|
|
VR 589 |
Potato |
0.01 |
(*) |
|
|
|
VC 431 |
Squash, Summer |
0.01 |
(*) |
|
|
|
TN 678 |
Walnuts |
0.01 |
(*) |
|
|
|
VC 432 |
Watermelon |
0.01 |
(*) |
|
|
|
181 |
MYCLOBUTANIL |
|
|
|
|
|
FS 12 |
Stone fruits |
2 |
|
(a) |
|
|
FB 275 |
Strawberry |
1 |
|
|
|
|
187 |
CLETHODIM |
|
|
|
|
|
AL 1020 |
Alfalfa fodder |
10 |
|
|
|
|
VP 61 |
Beans, except broad bean and soya bean |
0.5 |
(*) |
|
|
|
AM 1051 |
Fodder beet |
0.1 |
(*) |
|
|
|
VA 381 |
Garlic |
0.5 |
|
|
|
|
VA 385 |
Onion, Bulb |
0.5 |
|
|
|
|
SO 697 |
Peanut |
5 |
|
|
|
|
VO 448 |
Tomato |
1 |
|
|
|
|
189 |
TEBUCONAZOLE |
|
|
|
|
|
FS 13 |
Cherries |
5 |
|
|
|
|
DF 269 |
Dried grapes (=currants, raisins and sultanas) |
3 |
|
|
|
|
197 |
FENBUCONAZOLE |
|
|
|
|
|
FS 240 |
Apricot |
0.5 |
|
|
|
|
GC 640 |
Barley |
0.2 |
|
|
|
|
AS 640 |
Barley straw and fodder, Dry |
3 |
|
|
|
|
MF 812 |
Cattle fat |
0.05 |
(*) |
|
|
|
MO 1280 |
Cattle kidney |
0.05 |
(*) |
|
|
|
MO 1281 |
Cattle liver |
0.05 |
|
|
|
|
MM 812 |
Cattle meat |
0.05 |
(*) |
|
|
|
ML 812 |
Cattle milk |
0.05 |
(*) |
|
|
|
PE 112 |
Eggs |
0.05 |
(*) |
|
|
|
FS 247 |
Peach |
0.5 |
|
|
|
|
PF 111 |
Poultry fats |
0.05 |
(*) |
|
|
|
PM 110 |
Poultry meat |
0.05 |
(*) |
|
|
|
PO 111 |
Poultry, Edible offal of |
0.05 |
(*) |
|
|
|
SO 495 |
Rape seed |
0.05 |
(*) |
|
|
|
198 |
AMINOMETHYLPHOSPHONIC ACID (AMPA) |
|
|
|
|
|
GC 645 |
Maize |
2 |
|
|
|
|
AS 645 |
Maize fodder |
5 |
|
|
|
|
AF 645 |
Maize forage |
2 |
|
|
|
|
21 |
DDT |
|
|
|
|
|
MM 95 |
Meat (from mammals other than marine mammals) |
5 |
(fat) |
(a)[43] |
|
|
|
Commodity |
MRL
(mg/kg)[44] |
||||
|
14 |
CHLORFENVINPHOS |
|
|
|||
|
VB 400 |
Broccoli |
0.05 |
|
|||
|
VS 624 |
Celery |
0.4 |
|
|||
|
FC 1 |
Citrus fruits |
1 |
|
|||
|
SO 691 |
Cotton seed |
0.05 |
|
|||
|
VO 440 |
Egg plant |
0.05 |
|
|||
|
VR 583 |
Horseradish |
0.1 |
|
|||
|
VA 384 |
Leek |
0.05 |
|
|||
|
GC 645 |
Maize |
0.05 |
|
|||
|
MM 95 |
|
Meat (from mammals other than marine mammals) |
0.2 |
(fat) V |
||
|
ML 107 |
Milk of cattle, goats & sheep |
0.008 |
F V |
|||
|
VO 450 |
Mushrooms |
0.05 |
|
|||
|
VA 385 |
Onion, Bulb |
0.05 |
|
|||
|
SO 697 |
Peanut |
0.05 |
|
|||
|
VR 589 |
Potato |
0.05 |
|
|||
|
VR 494 |
Radish |
0.1 |
|
|||
|
GC 649 |
Rice |
0.05 |
|
|||
|
CM 1205 |
|
Rice, Polished |
0.05 |
|
||
|
VR 497 |
Swede |
0.05 |
|
|||
|
VR 508 |
Sweet potato |
0.05 |
|
|||
|
VO 448 |
Tomato |
0.1 |
|
|||
|
VR 506 |
Turnip, Garden |
0.05 |
|
|||
|
GC 654 |
Wheat |
0.05 |
|
|||
|
26 |
DICOFOL |
|
|
|||
|
AO2 2 |
|
Fruits (except as otherwise listed) |
5 |
|
||
|
53 |
MEVINPHOS |
|
|
|||
|
FP 226 |
Apple |
0.5 |
|
|||
|
FS 240 |
Apricot |
0.2 |
|
|||
|
VR 577 |
Carrot |
0.1 |
|
|||
|
FS 13 |
Cherries |
1 |
|
|||
|
VL 480 |
Kale |
1 |
|
|||
|
VL 482 |
Lettuce, Head |
0.5 |
|
|||
|
VA 385 |
Onion, Bulb |
0.1 |
|
|||
|
FS 247 |
Peach |
0.5 |
|
|||
|
FP 230 |
Pear |
0.2 |
|
|||
|
VR 589 |
Potato |
0.1 |
|
|||
|
VR 506 |
Turnip, Garden |
0.1 |
|
|||
|
56 |
2-PHENYLPHENOL |
|
|
|||
|
FP 226 |
Apple |
25 |
Po |
|||
|
60 |
PHOSALONE |
|
|
|||
|
FC 1 |
Citrus fruits |
1 |
|
|||
|
FB 269 |
Grapes |
5 |
|
|||
|
VR 589 |
Potato |
0.1 |
(*) |
|||
|
65 |
THIABENDAZOLE |
|
|
|||
|
GC 80 |
Cereal grains |
0.2 |
|
|||
|
VA 385 |
Onion, Bulb |
0.1 |
|
|||
|
VR 596 |
Sugar beet |
5 |
|
|||
|
AV 596 |
Sugar beet leaves or tops |
10 |
|
|||
|
DM 596 |
Sugar beet molasses |
1 |
|
|||
|
AB 596 |
Sugar beet pulp, Dry |
5 |
|
|||
|
VO 448 |
Tomato |
2 |
|
|||
|
76 |
THIOMETON |
|
|
|||
|
FP 226 |
Apple |
0.5 |
|
|||
|
FS 240 |
Apricot |
0.5 |
|
|||
|
VB 41 |
Cabbages, Head |
0.5 |
|
|||
|
VR 577 |
Carrot |
0.05 |
(*) |
|||
|
VS 624 |
Celery |
0.5 |
|
|||
|
GC 80 |
Cereal grains |
0.05 |
(*) |
|||
|
FS 244 |
Cherry, Sweet |
0.5 |
|
|||
|
VL 469 |
Chicory leaves |
0.5 |
|
|||
|
VP 526 |
Common bean (pods and/or immature seeds) |
0.5 |
|
|||
|
OC 691 |
Cotton seed oil, Crude |
0.1 |
(*) |
|||
|
VO 440 |
Egg plant |
0.5 |
|
|||
|
VL 476 |
Endive |
0.5 |
|
|||
|
AM 1051 |
|
Fodder beet |
0.05 |
(*) |
||
|
AV 1051 |
|
Fodder beet leaves or tops |
0.05 |
(*) |
||
|
FB 269 |
Grapes |
0.5 |
|
|||
|
DH 1100 |
|
Hops, Dry |
2 |
|
||
|
VL 482 |
Lettuce, Head |
0.5 |
|
|||
|
AF 645 |
Maize forage |
0.1 |
(*) fresh wt |
|||
|
SO 90 |
Mustard seeds |
0.05 |
(*) |
|||
|
HH 740 |
Parsley |
0.5 |
|
|||
|
FS 247 |
Peach |
0.5 |
|
|||
|
SO 703 |
Peanut, Whole |
0.5 |
|
|||
|
FP 230 |
Pear |
0.5 |
|
|||
|
VP 63 |
Peas (pods and succulent=immature seeds) |
0.5 |
|
|||
|
VO 51 |
Peppers |
0.5 |
|
|||
|
FS 14 |
Plums (including prunes) |
0.5 |
|
|||
|
VR 589 |
Potato |
0.05 |
(*) |
|||
|
FP 231 |
Quince |
0.5 |
|
|||
|
SO 495 |
Rape seed |
0.05 |
(*) |
|||
|
AS 81 |
Straw and fodder (dry) of cereal grains |
0.1 |
(*) |
|||
|
FB 275 |
Strawberry |
0.5 |
|
|||
|
VR 596 |
Sugar beet |
0.05 |
(*) |
|||
|
AV 596 |
Sugar beet leaves or tops |
0.05 |
(*) |
|||
|
VO 448 |
Tomato |
0.5 |
|
|||
|
90 |
CHLORPYRIFOS-METHYL |
|
|
|||
|
GC 645 |
Maize |
10 |
Po |
|||
|
96 |
CARBOFURAN |
|
|
|||
|
GC 640 |
Barley |
0.1 |
(*) |
|||
|
VB 402 |
Brussels sprouts |
2 |
|
|||
|
VB 41 |
Cabbages, Head |
0.5 |
|
|||
|
VB 404 |
Cauliflower |
0.2 |
|
|||
|
DH 1100 |
Hops, Dry |
5 |
|
|||
|
VB 405 |
Kohlrabi |
0.1 |
(*) |
|||
|
VL 482 |
Lettuce, Head |
0.1 |
(*) |
|||
|
SO 90 |
Mustard seeds |
0.1 |
(*) |
|||
|
FS 247 |
Peach |
0.1 |
(*) |
|||
|
FP 230 |
Pear |
0.1 |
(*) |
|||
|
FB 275 |
Strawberry |
0.1 |
(*) |
|||
|
103 |
PHOSMET |
|
|
|||
|
FI 335 |
Feijoa |
2 |
|
|||
|
FI 341 |
Kiwifruit |
15 |
|
|||
|
112 |
PHORATE |
|
|
|||
|
GC 640 |
Barley |
0.05 |
|
|||
|
SO 495 |
Rape seed |
0.1 |
|
|||
|
VO 448 |
Tomato |
0.1 |
|
|||
|
114 |
GUAZATINE |
|
|
|||
|
GC 80 |
Cereal grains |
0.1 |
(*) |
|||
|
FC 1 |
Citrus fruits |
5 |
Po |
|||
|
VC 46 |
Melons, except watermelon |
5 |
Po |
|||
|
FI 353 |
Pineapple |
0.1 |
(*) |
|||
|
VR 589 |
Potato |
0.1 |
(*) |
|||
|
GS 659 |
Sugar cane |
0.1 |
(*) |
|||
|
117 |
ALDICARB |
|
|
|||
|
FI 0327 |
Banana |
0.5 |
|
|||
|
128 |
PHENTHOATE |
|
|
|||
|
MM 812 |
Cattle meat |
0.05 |
(*) |
|||
|
FC 1 |
Citrus fruits |
1 |
|
|||
|
PE 112 |
Eggs |
0.05 |
(*) |
|||
|
ML 106 |
Milks |
0.01 |
(*) |
|||
|
CM 649 |
Rice, Husked |
0.05 |
|
|||
|
141 |
PHOXIM |
|
|
|||
|
VB 403 |
Cabbage, Savoy |
0.05 |
(*) |
|||
|
MM 812 |
Cattle meat |
0.2 |
(fat) V |
|||
|
VB 404 |
Cauliflower |
0.05 |
(*) |
|||
|
GC 80 |
Cereal grains |
0.05 |
(*) |
|||
|
VP 526 |
Common bean (pods and/or immature seeds) |
0.05 |
(*) |
|||
|
SO 691 |
Cotton seed |
0.05 |
(*) |
|||
|
VL 482 |
Lettuce, Head |
0.1 |
|
|||
|
ML 106 |
Milks |
0.05 |
F V |
|||
|
VA 385 |
Onion, Bulb |
0.05 |
(*) |
|||
|
VR 589 |
Potato |
0.05 |
(*) |
|||
|
MM 822 |
Sheep meat |
0.5 |
(fat) V |
|||
|
VO 447 |
Sweet corn (corn-on-the-cob) |
0.05 |
(*) |
|||
|
VO 448 |
Tomato |
0.2 |
|
|||
|
|
Commodity |
MRL (mg/kg) |
|
|
8 |
CARBARYL |
|
|
|
AL 1021 |
Alfalfa forage (green) |
100 |
|
|
FP 226 |
Apple |
5 |
|
|
FS 240 |
Apricot |
10 |
|
|
VS 621 |
Asparagus |
10 |
|
|
FI 327 |
Banana |
5 |
|
|
GC 640 |
Barley |
5 |
Po |
|
AL 1030 |
Bean forage (green) |
100 |
|
|
VR 574 |
Beetroot |
2 |
|
|
FB 264 |
Blackberries |
10 |
|
|
FB 20 |
Blueberries |
7 |
|
|
VB 41 |
Cabbages, Head |
5 |
|
|
VR 577 |
Carrot |
2 |
|
|
MM 812 |
Cattle meat |
0.2 |
|
|
FS 13 |
Cherries |
10 |
|
|
FC 1 |
Citrus fruits |
7 |
|
|
AL 1023 |
Clover |
100 |
fresh wt |
|
VP 526 |
Common bean (pods and/or immature seeds) |
5 |
|
|
SO 691 |
Cotton seed |
1 |
|
|
VD 527 |
Cowpea (dry) |
1 |
|
|
FB 265 |
Cranberry |
7 |
|
|
VC 424 |
Cucumber |
3 |
|
|
FB 266 |
Dewberries (including boysenberry and loganberry) |
10 |
|
|
VO 440 |
Egg plant |
5 |
|
|
PE 112 |
Eggs |
0.5 |
|
|
MM 814 |
Goat meat |
0.2 |
|
|
FB 269 |
Grapes |
5 |
|
|
AS 162 |
Hay or fodder (dry) of grasses |
100 |
|
|
FI 341 |
Kiwifruit |
10 |
fresh wt |
|
VL 53 |
Leafy vegetables |
10 |
|
|
AF 645 |
Maize forage |
100 |
|
|
VC 46 |
Melons, except watermelon |
3 |
|
|
AO3 1 |
Milk products |
0.1 |
(*) |
|
ML 106 |
Milks |
0.1 |
(*) |
|
FS 245 |
Nectarine |
10 |
|
|
AO51900 |
Nuts (whole in shell) |
10 |
|
|
GC 647 |
Oats |
5 |
Po |
|
VO 442 |
Okra |
10 |
|
|
FT 305 |
Olives |
10 |
|
|
DM 305 |
Olives, Processed |
1 |
|
|
VR 588 |
Parsnip |
2 |
|
|
AL 528 |
Pea vines (green) |
100 |
fresh wt |
|
FS 247 |
Peach |
10 |
|
|
AL 697 |
Peanut fodder |
100 |
|
|
SO 703 |
Peanut, Whole |
2 |
|
|
AL 697 |
Peanut fodder |
100 |
|
|
FP 230 |
Pear |
5 |
|
|
VP 63 |
Peas (pods and succulent=immature seeds) |
5 |
|
|
VO 51 |
Peppers |
5 |
|
|
FS 14 |
Plums (including prunes) |
10 |
|
|
VR 589 |
Potato |
0.2 |
|
|
PM 110 |
Poultry meat |
0.5 |
V |
|
PO 113 |
Poultry skin |
5 |
V |
|
VC 429 |
Pumpkins |
3 |
|
|
VR 494 |
Radish |
2 |
|
|
FB 272 |
Raspberries, Red, Black |
10 |
|
|
GC 649 |
Rice |
5 |
PoP |
|
CM 649 |
Rice, Husked |
5 |
Po |
|
GC 650 |
Rye |
5 |
Po |
|
MM 822 |
Sheep meat |
0.2 |
|
|
GC 651 |
Sorghum |
10 |
Po |
|
AF 651 |
Sorghum forage (green) |
100 |
fresh wt |
|
VD 541 |
Soya bean (dry) |
1 |
|
|
AL 1265 |
Soya bean forage (green) |
100 |
fresh wt |
|
VC 431 |
Squash, Summer |
3 |
|
|
FB 275 |
Strawberry |
7 |
|
|
VR 596 |
Sugar beet |
0.2 |
|
|
AV 596 |
Sugar beet leaves or tops |
100 |
|
|
VR 497 |
Swede |
2 |
|
|
VO 1275 |
Sweet corn (kernels) |
1 |
|
|
VO 448 |
Tomato |
5 |
|
|
TN 85 |
Tree nuts |
1 |
|
|
GC 654 |
Wheat |
5 |
Po |
|
CM 654 |
Wheat bran, Unprocessed |
20 |
PoP |
|
CF 1211 |
Wheat flour |
0.2 |
PoP |
|
CF 1212 |
Wheat wholemeal |
2 |
PoP |
|
VC 433 |
Winter squash |
3 |
|
|
65 |
THIABENDAZOLE |
|
|
|
FI 327 |
Banana |
3 |
|
|
VR 589 |
Potato |
5 |
Po |
|
81 |
CHLOROTHALONIL |
|
|
|
FB 21 |
Currants, Black, Red, White |
25 |
|
|
FS 247 |
Peach |
25 |
|
|
96 |
CARBOFURAN |
|
|
|
AL 1020 |
Alfalfa fodder |
20 |
|
|
SB 716 |
Coffee beans |
0.1 |
(*) |
|
VR 589 |
Potato |
0.5 |
|
|
105 |
DITHIOCARBAMATES |
|
|
|
FP 226 |
Apple |
3 |
|
|
FI 327 |
Banana |
1 |
|
|
VR 577 |
Carrot |
0.5 |
|
|
VC 424 |
Cucumber |
0.5 |
|
|
FB 21 |
Currants, Black, Red, White |
5 |
|
|
VL 482 |
Lettuce, Head |
5 |
|
|
VC 46 |
Melons, except watermelon |
1 |
|
|
FP 230 |
Pear |
3 |
|
|
VR 589 |
Potato |
0.1 |
|
|
VO 448 |
Tomato |
3 |
|
|
GC 654 |
Wheat |
0.2 |
|
|
158 |
GLYPHOSATE |
|
|
|
SO 691 |
Cotton seed |
0.5 |
|
|
GC 645 |
Maize |
0.1 |
(*) |
|
GC 651 |
Sorghum |
0.1 |
(*) |
The following is the final or tentative lists of compounds to be considered by the FAO/WHO Joint Meeting of Pesticide Residues (JMPR) from 1999 - 2004 (as of 17 April 1999):
AGENDA OF THE 1999 JMPR
|
Toxicological evaluations |
Residue evaluations |
|
NEW COMPOUNDS |
NEW COMPOUNDS |
|
pyriproxyfen |
pyriproxyfen |
|
PERIODIC REEVALUATIONS |
PERIODIC REEVALUATIONS |
|
|
bitertanol (144) |
|
chlorpyrifos (017) |
|
|
dimethipin (151) |
|
|
ethoprophos (149) |
|
|
|
ethoxyquin (035) |
|
|
fenamiphos (085) |
|
|
malathion (049) |
|
|
methiocarb (132) |
|
2-phenylphenol (056) |
2-phenylphenol (056) |
|
permethrin (120) |
|
|
propargite (113) |
|
|
pyrethrins (063) |
|
|
EVALUATIONS |
EVALUATIONS |
|
|
buprofezin (173) |
|
|
clethodim (187) |
|
|
diazinon (022) |
|
|
dinocap (087) |
|
|
ethephon (106) |
|
|
fenpropimorph (188) |
|
|
fenpyroxymate (193) |
|
|
folpet (041) |
|
N-acetyl glufosinate (NAG) |
glufosinate ammonium (175) |
|
|
phosalone (060) |
|
PTU (150) |
|
|
Toxicological evaluations |
Residue evaluations |
|
NEW COMPOUNDS |
NEW COMPOUNDS |
|
chlorpropham |
|
|
|
fipronil |
|
PERIODIC REEVALUATIONS |
PERIODIC REEVALUATIONS |
|
acephate (95) |
|
|
|
amitraz (122) |
|
|
captan (007) |
|
|
chlorpyriphos (017) |
|
|
cypermethrin (118)* |
|
deltamethrin (135) |
|
|
|
diphenylamine (030) |
|
dodine (084) |
|
|
fenitrothion (037) |
|
|
imazalil (110) |
|
|
methamidiphos (100) |
|
|
|
parathion (058) |
|
|
parathion-methyl (059) |
|
|
piperonyl butoxide (62) |
|
|
pyrethrins (063) |
|
thiodicarb (154) |
|
|
vamidothion (078) |
|
|
EVALUATIONS |
EVALUATIONS |
|
|
aldicarb (117) |
|
carbaryl (008) |
|
|
|
chlorfenvinphos (14) |
|
chlormequat (015) - acute RfD |
chlormequat (15) |
|
DDT (21) |
DDT (21) |
|
|
fenthion (039) |
|
fipronil |
|
|
|
mevinphos (053) |
|
|
thiabendazole (065) |
TENTATIVE AGENDA OF THE 2001 JMPR
|
Toxicological evaluations |
Residue evaluations |
|
NEW COMPOUNDS |
NEW COMPOUNDS |
|
|
chlorpropham |
|
imidacloprid |
imidacloprid |
|
spinosad |
spinosad |
|
PERIODIC REEVALUATIONS |
PERIODIC REEVALUATIONS |
|
|
carbaryl (008) |
|
|
diflubenzuron (130) |
|
|
dimethipin (151) |
|
|
dodine (084) |
|
|
ethoprophos (149) |
|
|
fenitrothion (037) |
|
|
imazalil (110) |
|
lindane (048) |
|
|
mecarbam (124) |
|
|
|
methomyl (094)/thiodicarb (154) |
|
methoprene (147) |
|
|
oxamyl (126) |
|
|
|
permethrin (120) |
|
prochloraz (142) |
|
|
|
propargite (113) |
|
triazophos (143) |
|
|
EVALUATIONS |
EVALUATIONS |
|
diflubenzuron (130) |
|
|
|
diquat (031) |
|
guazatine (114) |
guazatine (114) |
|
methomyl (094) |
|
|
Toxicological evaluations |
Residue evaluations |
|
NEW COMPOUNDS |
NEW COMPOUNDS |
|
esfenvalerate* |
esfenvalerate* |
|
flutolanil |
flutolanil |
|
PERIODIC REEVALUATIONS |
PERIODIC REEVALUATIONS |
|
|
acephate (095) |
|
|
deltamethrin (135) |
|
|
methamidophos (100) |
|
metalaxyl-M** |
|
|
|
oxamyl (126) |
|
|
pirimiphos-methyl (086) |
|
|
procloraz (142) |
|
propamocarb (148) |
|
|
tolyfluanid (162) |
tolylfluanid (162) |
|
triadimefon (133) |
|
|
|
triazophos (143) |
|
|
vamidothion (078) |
|
EVALUATIONS |
EVALUATIONS |
|
carbofuran (096) |
carbofuran (096) |
|
|
dithiocarbamates (105) |
|
|
phosmet (103) |
** Whether it is a replacement chemical for metalaxyl needs to be confirmed
TENTATIVE AGENDA OF THE 2003 JMPR
|
Toxicological evaluations |
Residue evaluations |
|
NEW COMPOUNDS |
NEW COMPOUNDS |
|
quinclorac |
quinclorac |
|
PERIODIC REEVALUATIONS |
PERIODIC REEVALUATIONS |
|
bendiocarb (137) |
|
|
cyhexatin (067) |
cyhexatin (067) |
|
|
endosulfan (032) |
|
|
lindane (048) |
|
|
mecarbam (124) |
|
|
metalaxyl-M |
|
|
methoprene (147) |
|
|
propamocarb (148) |
|
|
propineb |
|
|
triadimefon (133) |
|
Toxicological evaluations |
Residue evaluations |
|
NEW COMPOUNDS |
NEW COMPOUNDS |
|
PERIODIC REEVALUATIONS |
PERIODIC REEVALUATIONS |
|
clofentezine (156) |
clofentesine (156) |
CANDIDATE COMPOUNDS FOR PERIODIC REVIEW
NOT YET SCHEDULED
|
anilazine2 |
|
benalaxyl2 |
|
cyhalothrin3 |
|
flucythrinate4 |
|
glyphosate1 |
|
metalaxyl3 |
|
paclobutrazol2 |
|
paraquat1 |
|
phorate1 |
|
pirimicarb4 |
|
procymidone2 |
|
propiconazole2 |
|
propoxur2 |
|
terbufos2 |
|
triforine (residues)4 |
2 New candidate compound for periodic review
3 Not supported for periodic reevaluation. However, there is support for MRLs based on the use of specific enantiomers/isomers
4 Awaiting scheduling date for review in the European Community
1. Candidates for EMRLs - Should EMRL estimation be restricted only to pesticides for which uses are no longer registered or approved by a national authority (completely banned, banned on foods, restricted uses)?
CCPR requests for JMPR estimation of EMRLs are to be limited to those pesticides (including metabolites, reaction products and accompanying contaminants of pesticide production and use) and pesticide-commodity combinations for which uses are no longer registered or approved at the national level for food/feed purposes, or for which the CCPR concludes that public health concerns have not been relieved in the absence of EMRLs. (Bolded text is intended to allow the CCPR, as a risk management decision option, to request JMPR estimation of EMRLs in cases where one or two countries insist on continued use of a pesticide which has been banned in most countries.)
2. Environmental Persistence - Should EMRLs be estimated only for chemicals which are persistent in the environment? If so, by what measure should that be defined?
EMRLS should be estimated only for discontinued pesticides (as defined in 1 above) which are persistent in the environment. It is suggested as a guide that EMRL consideration be given to former pesticides for which it is anticipated that residual residues of regulatory concern will likely occur for a period of 3 or more years after discontinuation of the use (under ideal conditions approximately the minimum time for an EMRL candidate to be scheduled, reviewed, recommended and adopted).
3. Residues in food/feed - Should residues need first to be found at some level of regulatory significance in foods/feeds in trade? If so, what kinds of measures of regulatory significance should be considered?
Yes, residues of regulatory significance should be occurring in food/feeds in trade. Measures of regulatory significance may include, but not be limited to, a potential health concern and/or other regulatory concerns such as environmental concerns which may be monitored with EMRLs in food/feed.
4. Trade issue - Should there be a trade problem (reported to CCPR) before EMRLs are estimated for a pesticide/commodity combination, or should it be based on the potential for a trade problem, because of its persistence and the presence of measurable residues.
Either condition is a basis for considering a request for JMPR EMRL estimation. A greater burden of proof would be expected when no trade problem has been occurring, perhaps based on a history of monitoring data.
5. Proof of source of residues - Should the country requesting EMRL estimation be requested/required to provide proof or some credible evidence or rationale that reported residues (the data base) are not the result of purposeful uses?
Yes, the requesting country has an obligation to provide some credible assurance that residues in a data base to support EMRL estimates do not result from intentional use. For example, in some cases it may be possible to document when a national use(s) was discontinued and/or in some cases possibly that monitoring data show residues are less than the norm when the use was approved.
6. Health aspect - related to 3 above, should a health risk concern be a requirement or the only basis or requesting EMRL estimations? This relates to consistency with WTO SPS[45] consistency concerns that have been expressed.
A potential health concern (e.g., possibility of intake exceeding the ADI) may be a major reason for requesting estimation of EMRLs. However, as described in 3 above and in more detail below under “periodic review”, other reasons may also qualify. For example, just the lack of a trading standard can create trade problems, even if there is no health concern per se. As long as EMRLs are not established so low as to create significant trade barriers, the Committee does not consider concerns other than “ADI exceedances” as a basis for requesting an EMRL estimate inconsistent with WTO SPS principles.
7. CCPR Priorities - Should the CCPR use the same criteria for scheduling JMPR review as used for MRLs? If not, how should it differ?
If the CCPR decides to develop criteria for EMRLs this question will need to be referred to a working Group on Priorities or to whatever working group is formed to develop the criteria.
8. Periodic reviews of EMRLs - Should the CCPR support a periodic JMPR re-evaluation of EMRLs?
The CCPR supports the concept of a periodic review of EMRLs with reevaluations approximately every 5 years if it can be scheduled. In no case should it be greater than 8 years. While a periodic review of all EMRLs for given chemicals at regular intervals is recommended, reconsideration of individual EMRLs may be considered outside a periodic review if extraordinary circumstances require it. If the EMRLs do not significantly restrict trade the CCPR does not consider periodic review of EMRLs inconsistent with WTO SPS principles.
9. CCPR Data Issue - Location/amount - Should the CCPR specify that a minimum data base (e.g., minimum number of countries, minimum number of samples/commodity?) be committed before requesting the JMPR to conduct EMRL estimates or should a request and data commitment from one country with a problem/concern suffice (meaning if other countries have a different opinion on the need they have the opportunity to submit data to support their view)?
The CCPR should leave to the JMPR whether data are adequate to make an EMRL estimate. The country making a request normally has a valid reason from its perspective for doing so. It has an obligation to provide good supporting documentation and the CCPR has an obligation to consider its concern, whether other countries have a similar problem or not.
JMPR RISK ASSESSMENT ISSUES
10. Types of data - What types of data should be provided for estimation of EMRLs? - The same toxicology data requirements as for MRLs? Routine random monitoring data? targeted monitoring data? Multi-year monitoring data? Other than toxicology and residue data what other data should data submitters be requested to provide, the same as for MRLs or just those related to the integrity of the residue data (e.g., analytical methodology, storage stability, sampling)?
The CCPR supports the JMPR practice of estimating EMRLs on the basis of random monitoring data. It does not consider “targeted” monitoring data in most cases to be appropriate for that purpose, recognising that it is useful for other purposes, including possible development of residue mitigation strategies.
If the CCPR concludes as a risk management decision, that a JMPR EMRL estimate based on random monitoring data would result in an unacceptable economic disruption and is convinced that residue reduction strategies are not possible, it may request the JMPR to consider re-estimating an EMRL on the basis of other than random monitoring data and request a new risk assessment based on that new estimate.
The CCPR leaves other data type requirement issues to the JMPR.
11. Standard format - Should data submissions be
required or requested to be in a standard format?
If so, which formats should be recommended?
The CCPR recommends that the JMPR specify what standard format they would prefer for EMRL data submissions if something more than guidance in the FAO Manual is needed.
12. Statistical treatment - Should data submitters be required or requested to provide a statistical treatment of the data in addition to the “raw” data provided? If so, what information should be requested (e.g., number of samples analyzed, number of samples found with residues, number within residue ranges, number with no detections (what level?), limit of detection/determination, percentile figures)?
Countries requesting EMRL estimations should be referred to JMPR general guidance for EMRLs provided in the 1997 FAO Manual on the Submission and Evaluation of Pesticide Residues Data for the Estimation of Maximum Residue Levels in Food and Feed. They should be encouraged to provide all of the information listed above, but it should be required only if the JMPR requires it.
13. Limits of Determination - It has been recommended that the JMPR should continue to recommend suitable limits of determination for EMRLs.
The CCPR supports that recommendation as long as there is a reasonable expectation that residues may occur in a given commodity (or commodity group). Revocation of EMRLs should be considered, once there is no longer evidence that residues are likely to occur in practice.
14. Commodity Group EMRLs - One country recommends that EMRLs be estimated for commodity groups where possible.
The Committee endorses this recommendation.
15. Outliers - The JMPR and some countries do not consider the frequently used term to be appropriate for EMRL situations. Some prefer the term “extreme values”.
The CCPR accepts the principle that there may be sound reasons for excluding extreme values when estimating an EMRL. The CCPR accepts that the JMPR will need to determine inclusion or exclusion of extreme values on a case-by-case basis and that in accordance with 10 above, under certain circumstances the CCPR may request the JMPR to consider the use of data other than random monitoring data. The CCPR recognizes that the JMPR must retain the flexibility to consider various factors or approaches for exclusion of extreme values (including percentile approaches, violation rates or others) according to the circumstances of a given data base. The CCPR recognizes the need for such judgements to be based on sound science and for the scientific and other bases to be well documented.
16. Violation rates - The JMPR has described its practice of using likely violation rates as one tool for helping it arrive at an EMRL recommendation. It assumes 0.5 to 1% violation rates would be unacceptable to most countries, but invites countries to express a view on this topic.
The Committee accepts that any EMRL must be protective of the public health in the first instance. Once this criteria is fully met the Committee supports analyzes which result in EMRLs which are not so low as to be readily indistinguishable from background and not so low as to result in unnecessary trade disruption. At the same time the Committee supports EMRLs that are not so high that they will not detect continued uses of discontinued products, localised hot spots or do not reflect evidence of the expected continued decline of contaminants resulting from former pesticide uses.
The CCPR recognizes that an arbitrary violation rate or range
would not be applicable to every situation. However, the CCPR accepts that the
taking into account by the JMPR of violation rates which are consistent with
actual enforcement practices of importing countries can be a useful tool to
augment other tools in the exclusion of extreme values in submitted monitoring
data. The CCPR recommends that the JMPR, in applying any violation rates to the
setting of EMRLs, document the scientific and other bases on which the violation
rate was set, in accord with CAC decisions and taking into account relevant WTO
rulings. The CCPR encourages countries to submit violation rates with respect to
incidences of trade disruption.